Science: Scientific Notation and Order of Magnitude Estimation
Using powers of ten to describe and estimate scientific quantities
Using powers of ten to describe and estimate scientific quantities
Science - Grade 9-12
- 1
A wavelength of blue light is about 0.00000042 meters. Write this length in scientific notation.
- 2
Avogadro's number is about 6.02 x 10^23 particles per mole. Write this number in standard notation using digits.
- 3
Sunlight takes about 500 seconds to reach Earth. If light travels at 3.0 x 10^8 meters per second, estimate the distance from the Sun to Earth.
- 4
A proton has a mass of about 1.67 x 10^-27 kilograms. An electron has a mass of about 9.11 x 10^-31 kilograms. About how many times more massive is a proton than an electron?
- 5
A force is measured as 7.8 x 10^5 newtons. To the nearest power of ten, what is its order of magnitude?
- 6
Compare 2.5 x 10^-4 grams and 8.0 x 10^-5 grams. Which mass is larger, and by what factor?
- 7
A classroom shelf has 40 textbooks, and each textbook has about 300 pages. Estimate the total number of pages in scientific notation and give its order of magnitude.
- 8
A molecule is about 1.3 x 10^-9 meters wide. Since 1 nanometer equals 1 x 10^-9 meters, write the molecule's width in nanometers.
- 9
A robot travels 4.2 x 10^6 meters, then travels another 7.5 x 10^5 meters. What total distance did it travel in scientific notation?
- 10
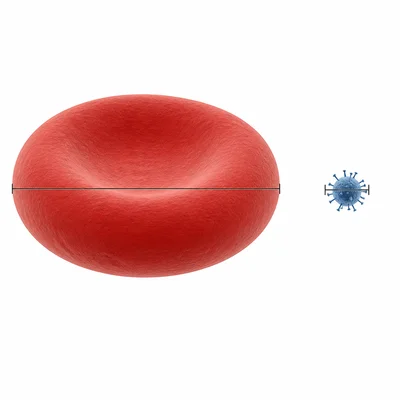
A red blood cell is about 8 x 10^-6 meters across, and a virus is about 1 x 10^-7 meters across. About how many times wider is the red blood cell than the virus?
- 11
The mass of a small asteroid is estimated to be 3 x 10^12 kilograms. The mass of a spacecraft is about 2 x 10^5 kilograms. About how many times more massive is the asteroid than the spacecraft?
- 12
Estimate the number of seconds in one year using 365 days per year, 24 hours per day, and 3600 seconds per hour. Write your answer in scientific notation.
- 13
A solution has a concentration of 0.00035 moles per liter. Write the concentration in scientific notation, then give the nearest power of ten.
- 14
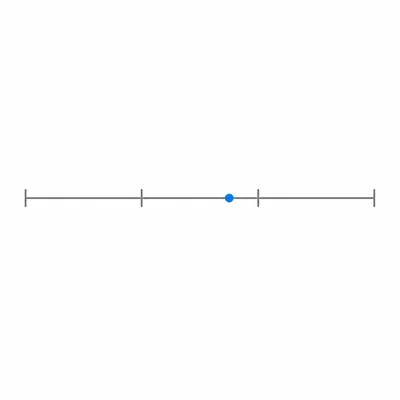
On a logarithmic scale marked 10^-3, 10^-2, 10^-1, and 10^0, place 5 x 10^-2 in the correct interval and state which power of ten it is closer to.
- 15
A small drop of water has a mass of about 0.05 grams. Water has a molar mass of about 18 grams per mole, and 1 mole contains about 6 x 10^23 molecules. Estimate the number of water molecules in the drop.
More Science Worksheets
Science: The Scientific Method
Grade 6-8 · 12 problems
Science: Food Chains and Food Webs
Grade 2-3 · 15 problems
Life Cycle of a Butterfly
Grade 2-3 · 15 problems
Science: Life Cycle of a Frog
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems