Stoichiometry Roadmap
Moles, Molar Mass, and Reaction Calculations
Related Tools
Related Labs
Related Worksheets
Related Cheat Sheets
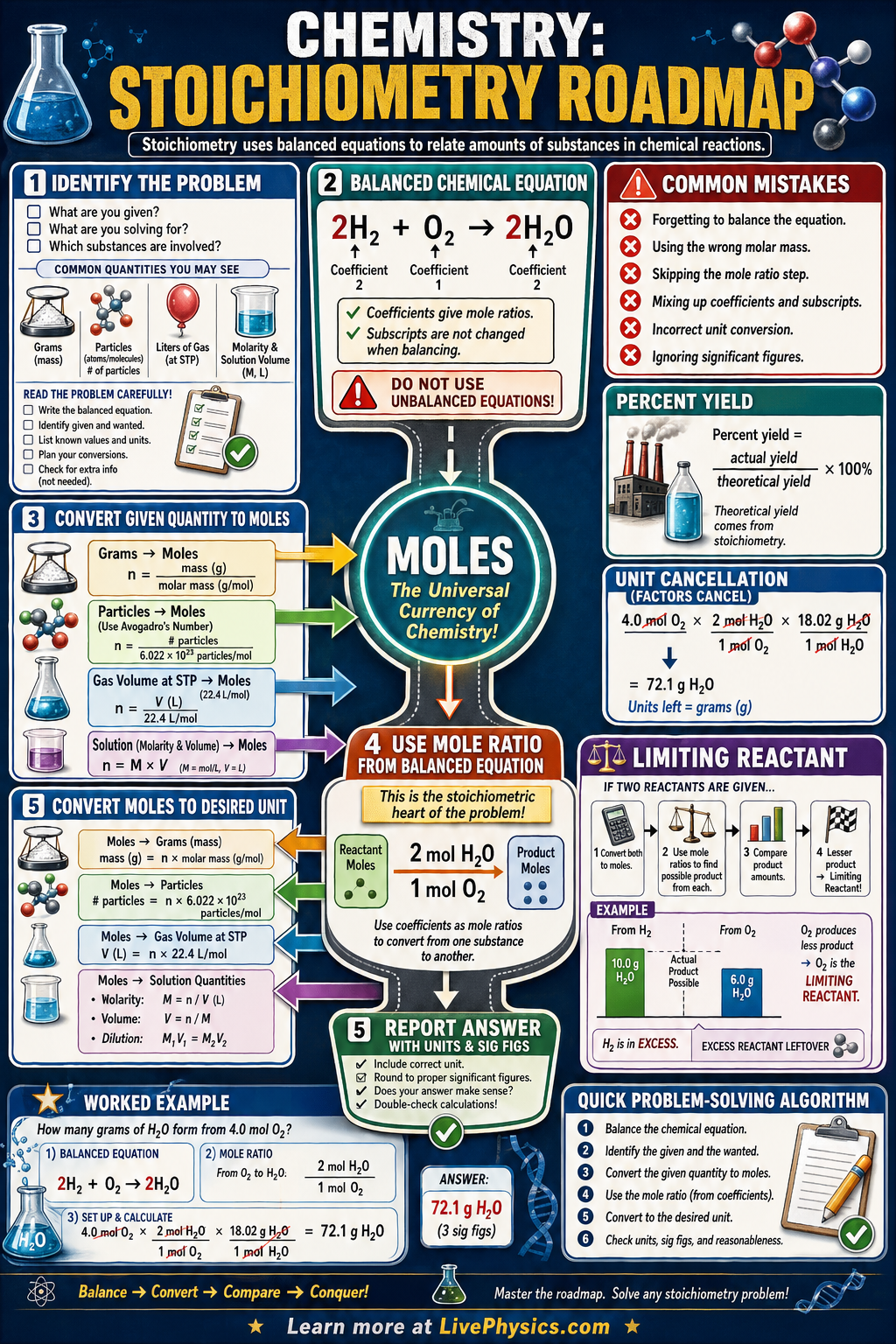
Stoichiometry is the branch of chemistry that uses balanced chemical equations to calculate the amounts of reactants consumed and products formed. The central unit is the mole: 1 mol = 6.022 × 10²³ particles (Avogadro's number). Converting between grams and moles requires the molar mass (grams per mole), obtained by summing atomic masses. Converting between moles of different substances requires the mole ratio from the balanced equation.
A typical stoichiometry problem follows a roadmap: grams of substance A → moles of A (divide by molar mass) → moles of B (multiply by mole ratio from equation) → grams of B (multiply by molar mass of B). Limiting reagent problems add a step: compute predicted moles of product from each reactant separately, then use the smaller value. The actual yield divided by the theoretical yield gives percent yield.
Key Facts
- 1 mol = 6.022 × 10²³ particles (Avogadro's number)
- Molar mass (g/mol) = of atomic masses of all atoms in the formula
- Mole ratio from balanced equation connects moles of one substance to another
- Stoichiometry map: grams A → moles A → moles B → grams B
- Limiting reagent: the reactant that runs out first and limits the amount of product
- Percent yield = (actual yield / theoretical yield) × 100%
Vocabulary
- Mole
- The SI unit for amount of substance; 1 mol = 6.022 × 10²³ representative particles (atoms, ions, or molecules).
- Molar mass
- The mass of one mole of a substance in grams per mole; numerically equal to the formula mass in atomic mass units.
- Limiting reagent
- The reactant that is completely consumed first in a reaction, limiting the amount of product that can form.
- Theoretical yield
- The maximum amount of product that can form from the given amounts of reactants, calculated from stoichiometry.
- Percent yield
- The ratio of actual yield to theoretical yield, expressed as a percentage; measures efficiency of a reaction.
Common Mistakes to Avoid
- Skipping the mole ratio step and directly converting grams of reactant to grams of product by mass ratio. The mass ratio between reactant and product is not the same as the mole ratio - you must go through moles and use the balanced equation.
- Using an unbalanced equation for stoichiometry. Mole ratios only have meaning from a balanced equation. Always balance before calculating.
- Forgetting to identify the limiting reagent in problems with two reactants. The reagent present in excess does not determine how much product forms.
- Computing percent yield greater than 100% without questioning it. A percent yield above 100% indicates experimental error - typically incomplete drying, impurities, or a measurement mistake.
Practice Questions
- 1 How many grams of H₂O form when 4.0 g of H₂ reacts with excess O₂? (Balanced: 2H₂ + O₂ → 2H₂O)
- 2 In the reaction N₂ + 3H₂ → 2NH₃, if 14 g of N₂ and 6 g of H₂ are mixed, which is the limiting reagent and how many grams of NH₃ are produced?
- 3 A student synthesizes 4.2 g of aspirin (theoretical yield 5.0 g). What is the percent yield?