Science: Atoms Elements and the Periodic Table
Exploring atomic structure, elements, and how the periodic table is organized
Exploring atomic structure, elements, and how the periodic table is organized
Chemistry - Grade 6-8
- 1
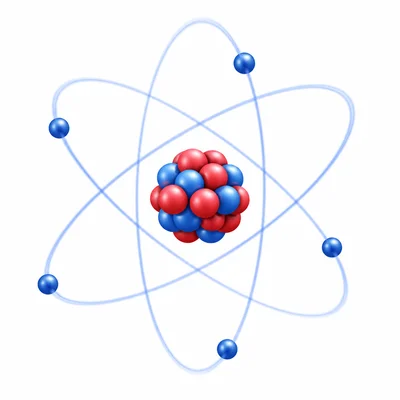
Name the three main subatomic particles found in atoms and give the charge of each one.
- 2
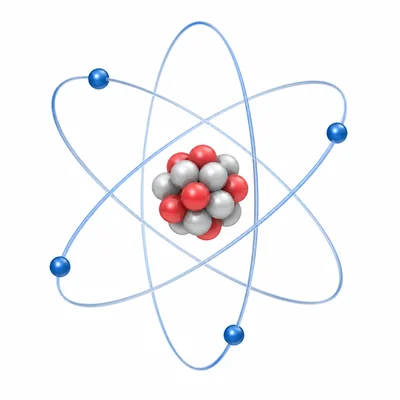
Where are protons, neutrons, and electrons located in an atom?
- 3
An atom has 11 protons. What element is it, and how do you know?
- 4
What does the atomic number of an element tell you?
- 5
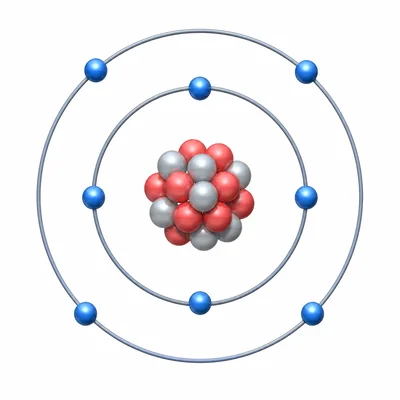
A neutral atom of oxygen has an atomic number of 8 and a mass number of 16. How many protons, neutrons, and electrons does it have?
- 6
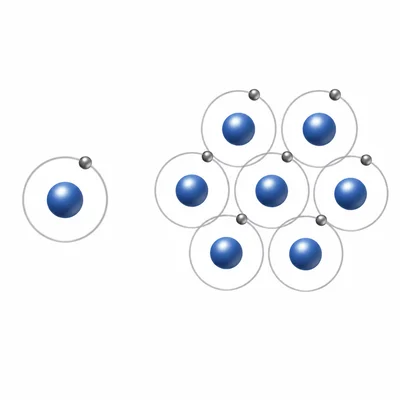
Explain the difference between an atom and an element.
- 7
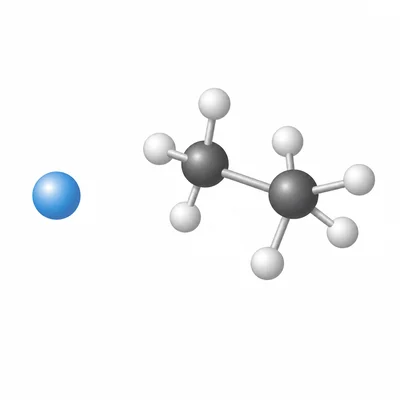
What is a molecule, and how is it different from a single atom?
- 8
Why is the periodic table useful to scientists and students?
- 9
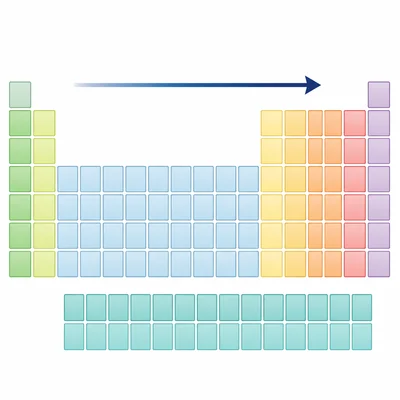
How are elements arranged across the periodic table?
- 10
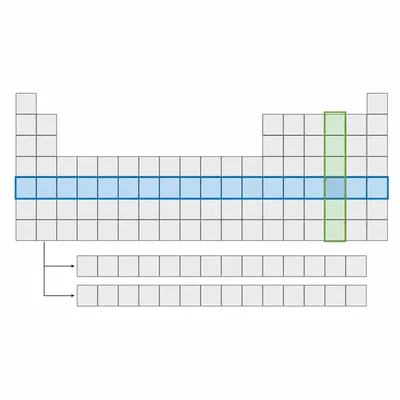
What is the difference between a period and a group on the periodic table?
- 11
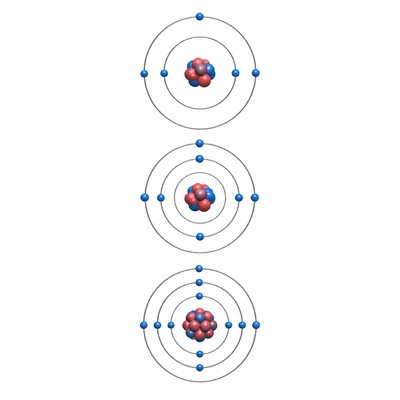
Elements in the same group often behave in similar ways. Give one reason why.
- 12
Classify each element as a metal, nonmetal, or metalloid: iron, oxygen, silicon.
- 13
A neutral atom has 17 electrons. What is its atomic number, and what element is it?
- 14
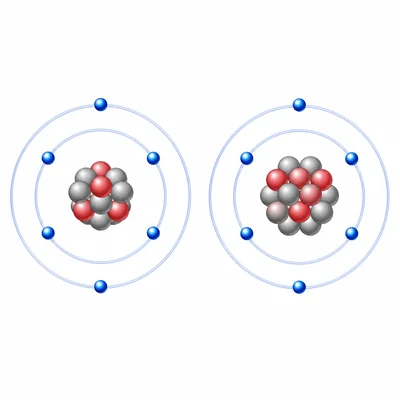
Compare the atoms of carbon and carbon-14. How are they similar and how are they different?
- 15
A student says that all squares on the periodic table represent compounds. Explain why this statement is incorrect.
Related Tools
Related Labs
Related Infographics
Related Mission Packs
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
Science: Periodic Table Trends
Grade 9-12 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems