Physics: AP Physics 2: Thermodynamics and Heat Engines
Work, heat, internal energy, entropy, and engine efficiency
Work, heat, internal energy, entropy, and engine efficiency
Physics - Grade 9-12
- 1
A gas absorbs 500 J of heat from its surroundings and does 200 J of work on the surroundings. What is the change in the gas's internal energy?
- 2
An ideal gas is compressed in a cylinder. During the compression, 150 J of work is done on the gas, and the gas releases 60 J of heat to the surroundings. What is the change in internal energy of the gas?
- 3
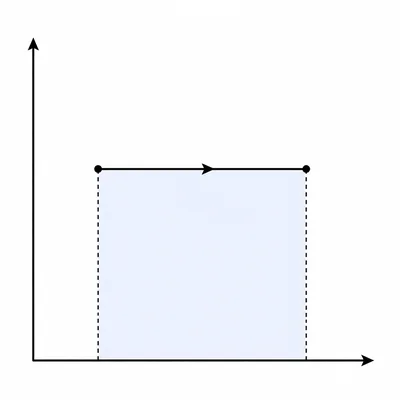
A gas expands at a constant pressure of 2.0 x 10^5 Pa from a volume of 0.010 m^3 to 0.030 m^3. How much work does the gas do?
- 4
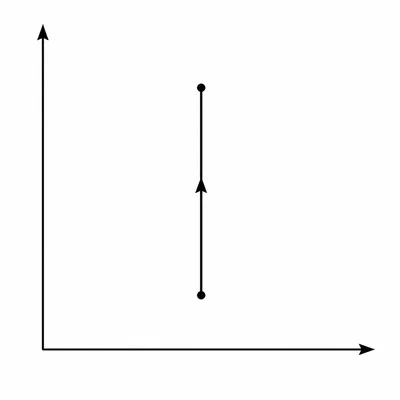
An ideal gas undergoes an isochoric process. Its pressure increases while its volume remains constant. Explain why the work done by the gas is zero.
- 5
A sample of ideal gas is heated at constant volume. The gas absorbs 240 J of heat. How much work is done by the gas, and what is the change in internal energy?
- 6
A heat engine absorbs 1200 J of energy from a hot reservoir and exhausts 750 J of energy to a cold reservoir during each cycle. How much work does the engine do per cycle, and what is its efficiency?
- 7
A Carnot engine operates between a hot reservoir at 600 K and a cold reservoir at 300 K. What is the maximum possible efficiency of the engine?
- 8
A student claims that a heat engine operating between 500 K and 250 K can have an efficiency of 60%. Is this possible? Explain your answer.
- 9
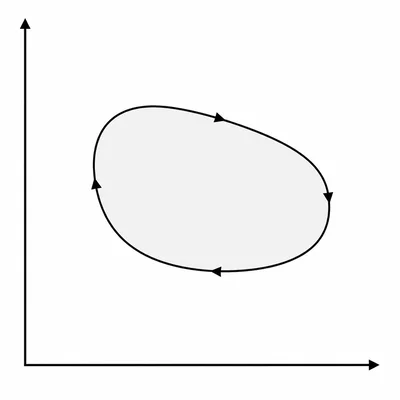
A cyclic process is shown on a PV diagram as a closed clockwise loop. Explain whether the net work done by the gas over one cycle is positive, negative, or zero.
- 10
A refrigerator removes 300 J of heat from its cold interior while 100 J of work is done on it. How much heat is expelled to the warmer room, and what is the refrigerator's coefficient of performance?
- 11
During an irreversible process, 600 J of heat flows from a hot object at 400 K to a cold object at 300 K. Calculate the total entropy change of the two-object system.
- 12
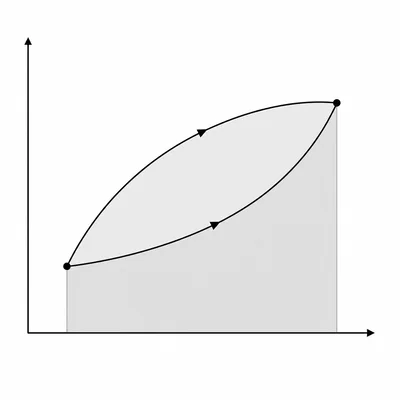
A PV diagram shows an ideal gas moving from state A to state B along two different paths. Path 1 has a larger area under the curve than Path 2. Compare the work done by the gas along the two paths, and explain whether the change in internal energy depends on the path.
More Physics Worksheets
Forces & Motion
Grade 6-8 · 8 problems
Science: Energy Thermodynamics and Heat Transfer
Grade 9-12 · 15 problems
Science: Light and Sound
Grade 4-5 · 15 problems
Science: Newtons Laws of Motion
Grade 9-12 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems