Science: Energy Thermodynamics and Heat Transfer
Thermal energy, the laws of thermodynamics, and mechanisms of heat transfer
Thermal energy, the laws of thermodynamics, and mechanisms of heat transfer
Physics - Grade 9-12
- 1

Define temperature and thermal energy. Then explain how they are related but not the same.
- 2
State the first law of thermodynamics in your own words and give one everyday example that shows this law.
- 3
A 2.0 kg block of metal is heated and absorbs 9000 J of energy. If the block's temperature increases by 15 degrees C, what is its specific heat capacity?
- 4
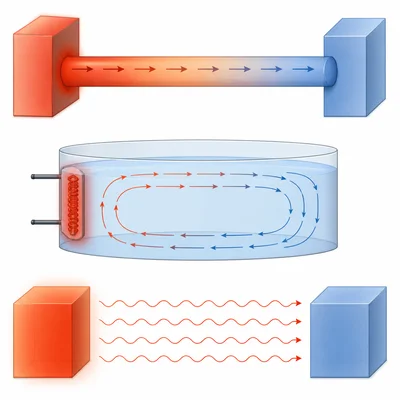
Explain the difference between conduction, convection, and radiation. Give one example of each.
- 5
A 0.50 kg sample of water cools from 80 degrees C to 30 degrees C. How much thermal energy does it lose? Use c = 4186 J/kg degrees C.
- 6

Describe thermal equilibrium. What happens to net heat flow when two objects reach thermal equilibrium?
- 7
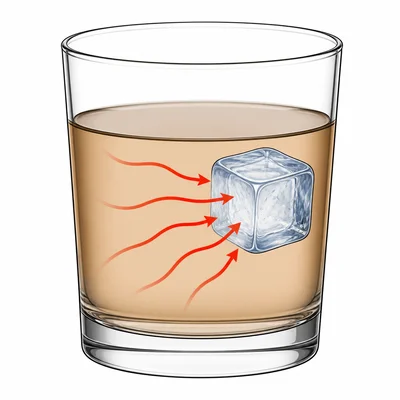
A student says, "Cold flows from the ice cube into the drink." Explain what is wrong with this statement.
- 8
What does the second law of thermodynamics say about the direction of natural processes and energy transfers?
- 9
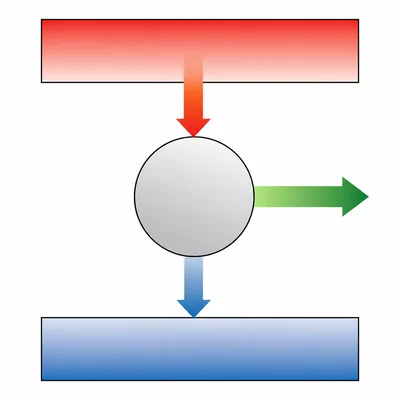
A heat engine takes in 1200 J of energy from a hot source and releases 750 J to a cold sink. How much work does the engine do?
- 10
Calculate the efficiency of the heat engine in the previous problem.
- 11
Why can no heat engine be 100 percent efficient according to the second law of thermodynamics?
- 12
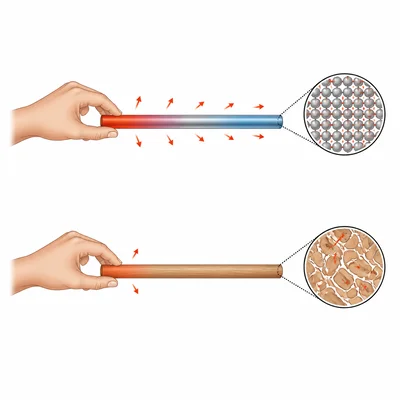
A copper rod and a wooden rod are left in the same room overnight. In the morning they have the same temperature, but the copper rod feels colder. Explain why.
- 13
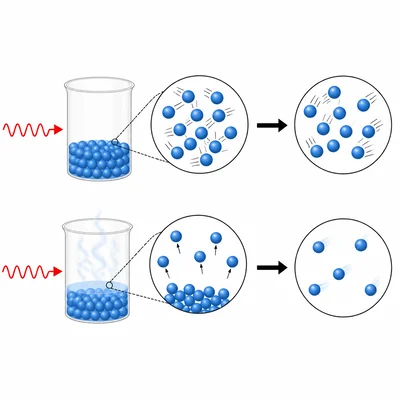
During a phase change such as melting or boiling, why can a substance absorb energy without increasing in temperature?
- 14
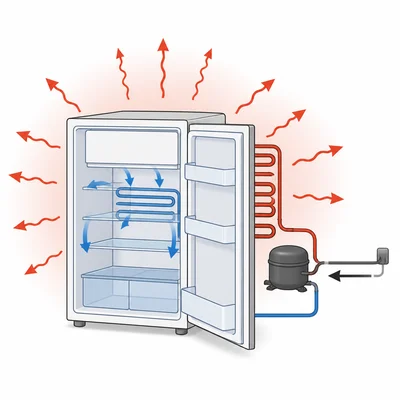
A refrigerator removes 500 J of thermal energy from its inside and releases 650 J to the room. How much work does the refrigerator require?
- 15
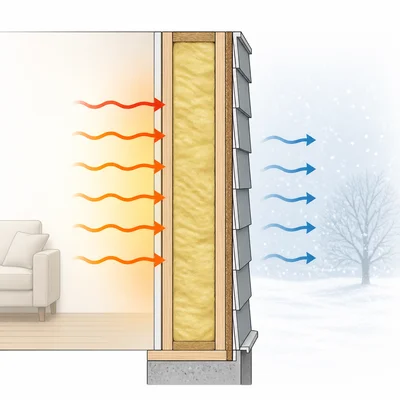
Name one way insulation reduces heat transfer in a house, and identify which method or methods of heat transfer it mainly reduces.
Related Tools
Related Infographics
Related Cheat Sheets
More Physics Worksheets
Forces & Motion
Grade 6-8 · 8 problems
Science: Light and Sound
Grade 4-5 · 15 problems
Science: Newtons Laws of Motion
Grade 9-12 · 15 problems
Science: Simple Machines
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems