Chemistry: Heat, Energy, and Phase Changes
Exploring how heat affects matter and changes of state
Exploring how heat affects matter and changes of state
Chemistry - Grade 6-8
- 1

A student places an ice cube on a warm plate. Explain what happens to the ice cube and describe how energy moves during this change.
- 2

What is the difference between heat and temperature?
- 3
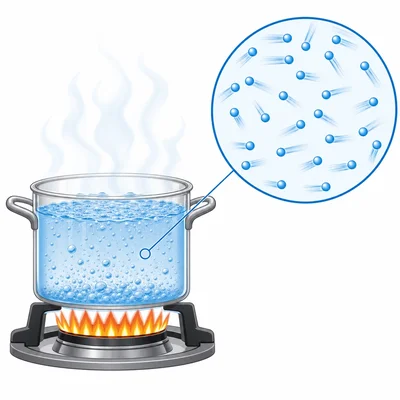
A pot of water is heated on a stove. The water temperature rises from 25 degrees Celsius to 100 degrees Celsius. What happens to the motion of the water particles as the temperature increases?
- 4

Name the phase change that occurs when liquid water becomes water vapor. Explain whether energy is absorbed or released.
- 5

Name the phase change that occurs when water vapor turns into liquid water on the outside of a cold glass. Explain why this happens.
- 6
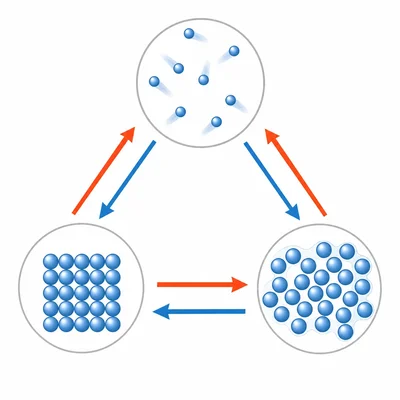
Sort these phase changes into two groups: melting, freezing, condensation, evaporation, sublimation, deposition. Which absorb energy, and which release energy?
- 7
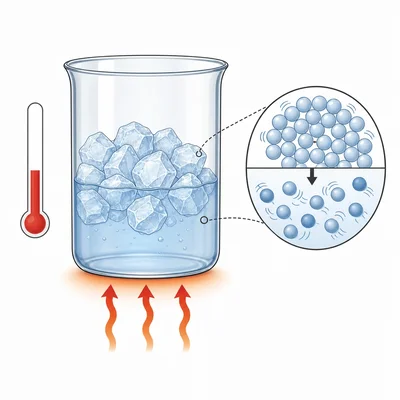
A solid substance is heated, but its temperature stays the same while it melts. Explain why the temperature does not increase during melting.
- 8
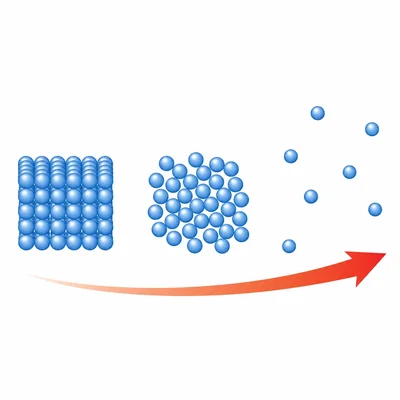
Use the terms solid, liquid, and gas to describe how particle arrangement changes as a substance gains energy.
- 9

Dry ice is solid carbon dioxide. At room temperature, it changes directly from a solid into a gas. What is this phase change called, and does it absorb or release energy?
- 10

Frost forms on a cold window when water vapor in the air changes directly into ice. What is this phase change called, and does it absorb or release energy?
- 11
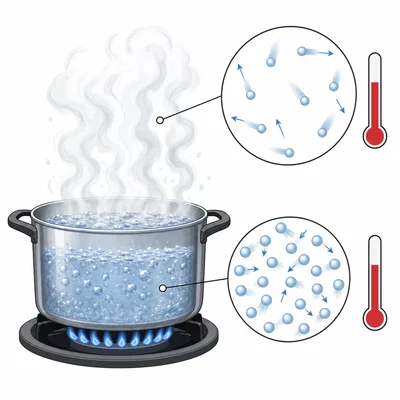
A student says, "Boiling water is hotter than steam because water is liquid and steam is gas." Explain why this statement can be incorrect.
- 12

A cup of hot chocolate is left on a table. After 20 minutes, it is cooler. Explain what happened to the thermal energy of the hot chocolate.
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems