Science: Chemical Reactions and Balancing Equations
Identify reaction evidence and balance simple chemical equations
Identify reaction evidence and balance simple chemical equations
Chemistry - Grade 6-8
- 1

List two common signs that a chemical reaction may have occurred.
- 2
Explain why subscripts in a chemical formula should not be changed when balancing an equation.
- 3
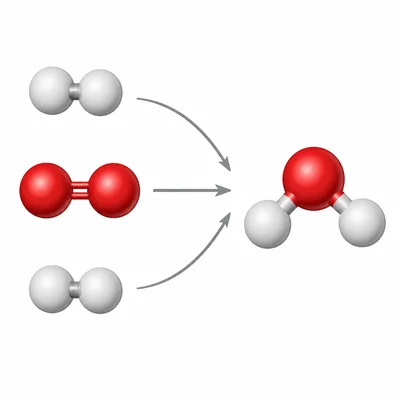
Balance the equation: H2 + O2 -> H2O
- 4
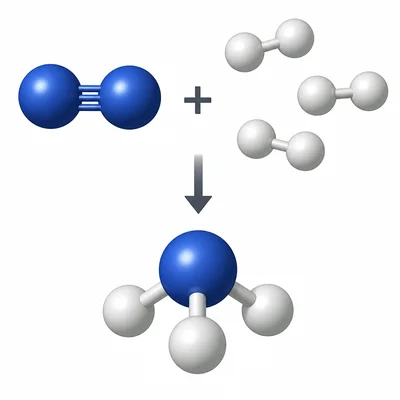
Balance the equation: N2 + H2 -> NH3
- 5
Balance the equation: Na + Cl2 -> NaCl
- 6
Balance the equation: Mg + O2 -> MgO
- 7
Balance the equation: Fe + O2 -> Fe2O3
- 8
Balance the equation: Al + O2 -> Al2O3
- 9
Balance the equation: Ca + H2O -> Ca(OH)2 + H2
- 10
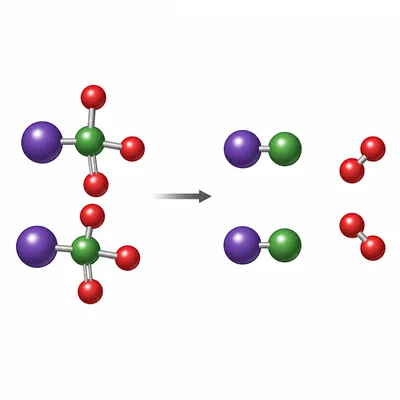
Balance the equation: KClO3 -> KCl + O2
- 11
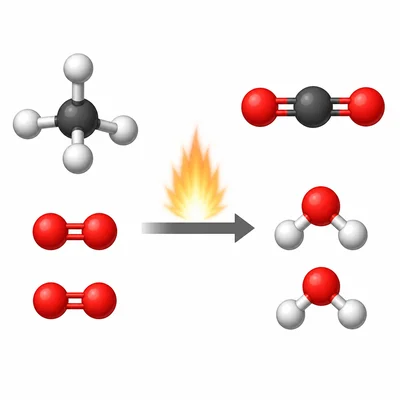
Balance the equation: CH4 + O2 -> CO2 + H2O
- 12
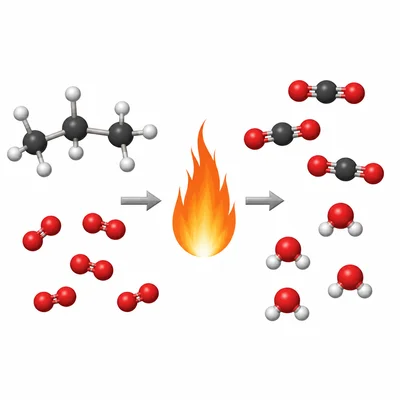
Balance the equation: C3H8 + O2 -> CO2 + H2O
- 13

A student says a balanced equation means the same number of molecules are on each side. Explain what is actually conserved in a balanced chemical equation.
- 14
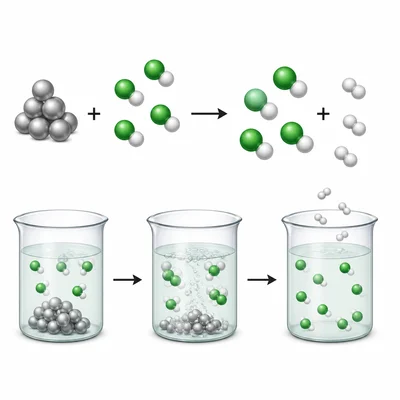
Balance the equation: Zn + HCl -> ZnCl2 + H2
- 15
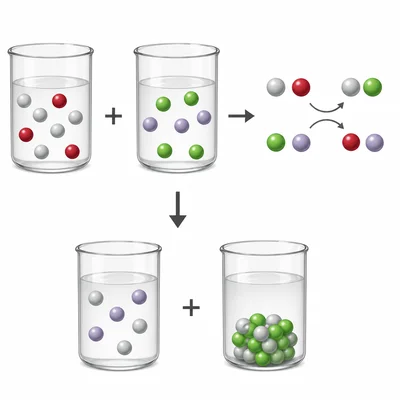
Balance the equation: AgNO3 + NaCl -> AgCl + NaNO3
Related Tools
Related Labs
Related Infographics
Related Mission Packs
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
Science: Periodic Table Trends
Grade 9-12 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems