Chemistry: Mixtures vs Solutions: Concentration and Saturation
Exploring solutes, solvents, concentration, and saturation
Exploring solutes, solvents, concentration, and saturation
Chemistry - Grade 6-8
- 1

Classify each example as a mixture or a solution: trail mix, salt water, muddy water, and lemonade with all sugar dissolved.
- 2

In a cup of sugar water, identify the solute and the solvent.
- 3
A student dissolves 10 grams of salt in 100 milliliters of water. What is the concentration in grams per milliliter?
- 4
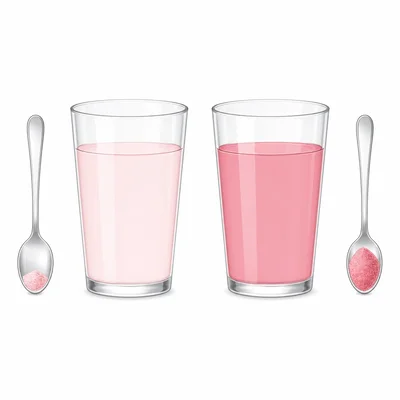
Two cups contain the same amount of water. Cup A has 1 teaspoon of drink mix dissolved in it. Cup B has 3 teaspoons of drink mix dissolved in it. Which cup has the higher concentration? Explain.
- 5

A solution contains 25 grams of sugar dissolved in 250 milliliters of water. A second solution contains 25 grams of sugar dissolved in 500 milliliters of water. Which solution is more dilute? Explain.
- 6

At room temperature, a glass of water can dissolve up to 36 grams of salt. If only 20 grams of salt are dissolved, is the solution saturated or unsaturated? Explain.
- 7
At room temperature, a glass of water can dissolve up to 36 grams of salt. If 36 grams are dissolved and no more salt can dissolve, what type of solution is it?
- 8

A student adds sugar to tea and sees sugar crystals sitting at the bottom after stirring for a long time. What does this show about the solution?
- 9

Explain how temperature can affect how much solid solute, such as sugar, dissolves in water.
- 10

A warm saturated sugar solution is carefully cooled and still contains more dissolved sugar than it normally could at the cooler temperature. What kind of solution is this?
- 11

Look at the data: Solution A has 5 grams of salt in 100 milliliters of water. Solution B has 15 grams of salt in 100 milliliters of water. Solution C has 15 grams of salt in 300 milliliters of water. Rank the solutions from most concentrated to least concentrated.
- 12

A diagram shows three beakers. Beaker 1 has clear salt water. Beaker 2 has sand and water with sand settled at the bottom. Beaker 3 has oil floating on water. Which beaker contains a solution, and how can you tell?
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems