Chemistry: States of Matter and Particle Models
Explaining solids, liquids, gases, and changes of state with particles
Explaining solids, liquids, gases, and changes of state with particles
Chemistry - Grade 6-8
- 1

A student draws particles in a solid as tightly packed circles in a fixed pattern. Explain what the particles are doing even though the solid keeps its shape.
- 2
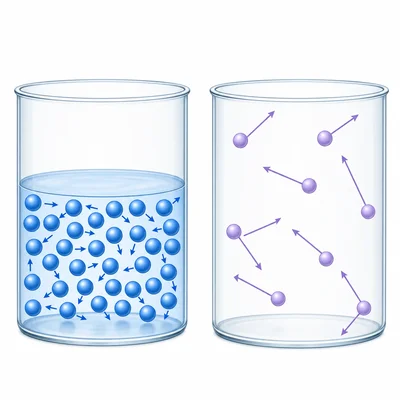
Compare the particle spacing and particle motion in a liquid and a gas.
- 3

A sealed balloon is placed in a warm room, and the balloon gets slightly larger. Use the particle model to explain why this happens.
- 4

Classify each substance as a solid, liquid, or gas at room temperature: ice cube, helium in a party balloon, cooking oil, and a metal spoon.
- 5

A beaker of water is heated on a hot plate. Describe what happens to the water particles as the temperature rises before boiling begins.
- 6

Name the change of state when water vapor cools and forms liquid droplets on the outside of a cold glass. Explain the particle change.
- 7
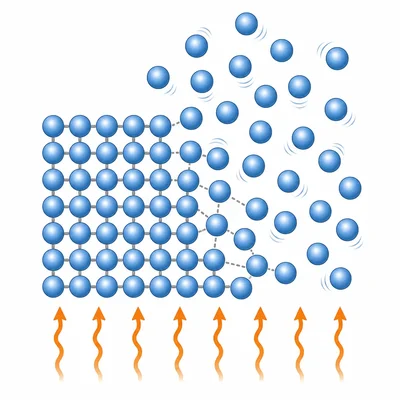
During melting, does the temperature of a pure substance usually keep rising, or does it stay nearly constant until melting is complete? Explain why.
- 8

A student says, "When a puddle evaporates, the water disappears and no longer exists." Correct this statement using the particle model.
- 9

Look at a particle diagram showing particles packed close together but arranged randomly and able to slide. Which state of matter does the diagram represent? Explain your answer.
- 10
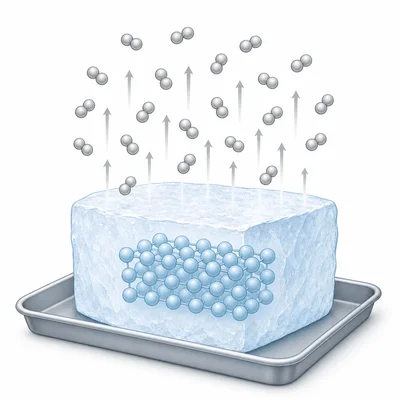
Dry ice is solid carbon dioxide. At room temperature, it changes directly from a solid to a gas without becoming a liquid. Name this change of state and describe the particle energy change.
- 11
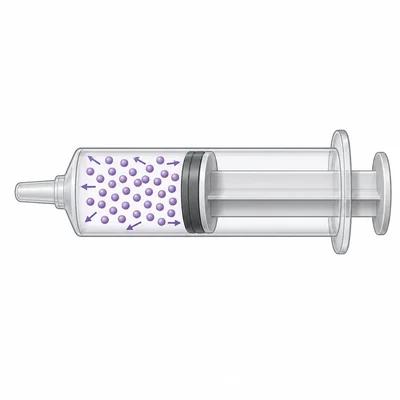
A gas is compressed in a syringe by pushing the plunger down while the tip is sealed. What happens to the spacing between gas particles and the volume of the gas?
- 12
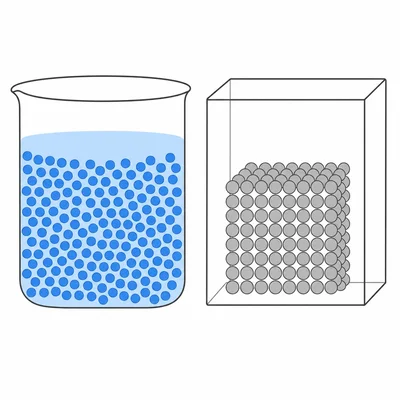
Explain why a liquid takes the shape of its container but a solid usually does not.
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems