Chemistry: Organic Chemistry: Polymerization Reactions
How monomers join to form addition and condensation polymers
How monomers join to form addition and condensation polymers
Chemistry - Grade 9-12
- 1
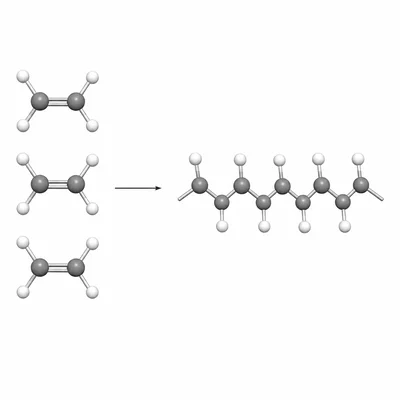
Ethene, CH2=CH2, can form polyethene through an addition polymerization reaction. Write the repeating unit of polyethene and explain what happens to the carbon-carbon double bond.
- 2
Classify the formation of polypropene from propene, CH2=CHCH3, as addition polymerization or condensation polymerization. Explain your choice.
- 3
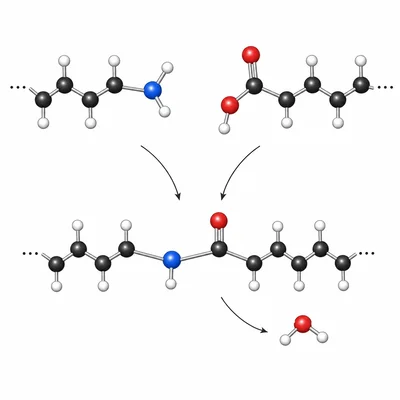
Nylon-6,6 forms when hexane-1,6-diamine reacts with hexanedioic acid. A small molecule is released each time an amide bond forms. Identify the type of polymerization and name the small molecule released.
- 4
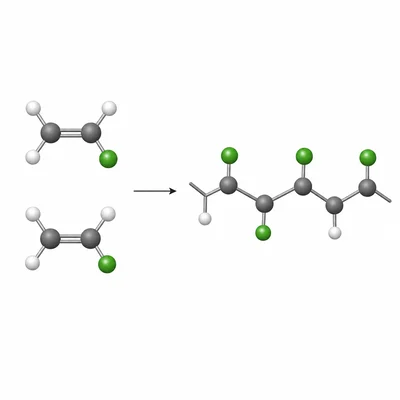
Vinyl chloride has the structure CH2=CHCl. Draw or describe the repeating unit of polyvinyl chloride, PVC.
- 5
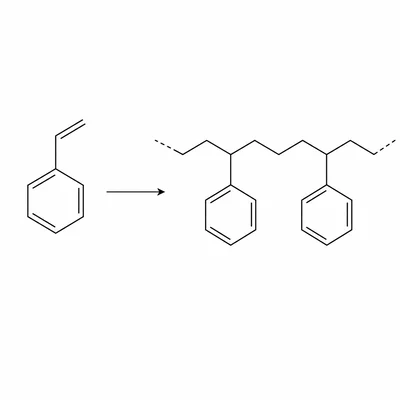
Explain the difference between a monomer and a polymer using styrene and polystyrene as examples.
- 6
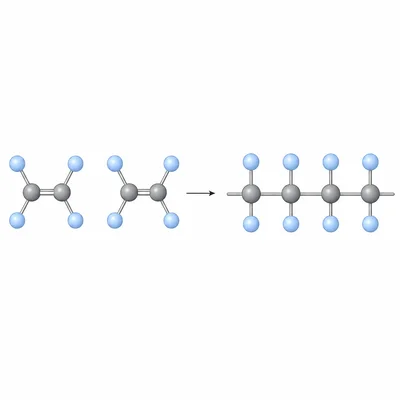
A polymer is formed from the monomer tetrafluoroethene, CF2=CF2. The product is polytetrafluoroethene, PTFE. Write the repeating unit and identify the polymerization type.
- 7
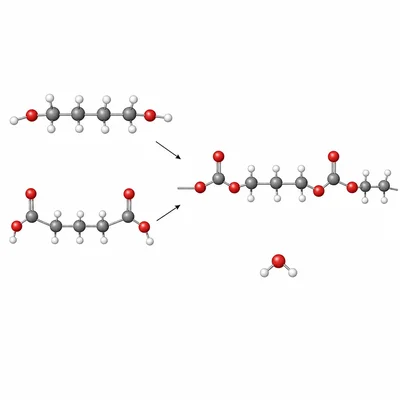
A polyester can form when a diol reacts with a dicarboxylic acid. What functional group forms in the polymer chain, and why is this reaction considered condensation polymerization?
- 8
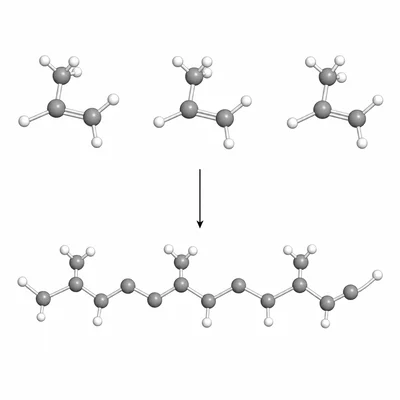
The diagram shows three propene monomers joining to begin a polypropene chain. Describe the repeating unit of polypropene in words or symbols.
- 9
Compare addition polymerization and condensation polymerization in terms of reactants and products.
- 10
A student says, "All polymerization reactions make water." Explain why this statement is incorrect.
- 11
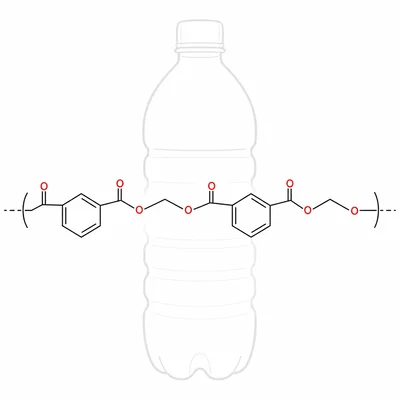
Polyethylene terephthalate, PET, is a polyester used in many plastic bottles. It forms from a diol and a dicarboxylic acid or related ester. Identify PET as an addition polymer or a condensation polymer and explain your reasoning.
- 12
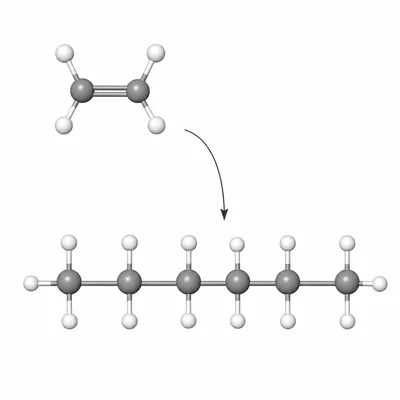
The image shows a section of a polymer chain with the repeating pattern -CH2-CH2-CH2-CH2-. Identify a possible monomer that could have formed this polymer by addition polymerization and explain your answer.
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems