Molecular Geometry and VSEPR
Shape, Bond Angles, and Polarity
Related Tools
Related Worksheets
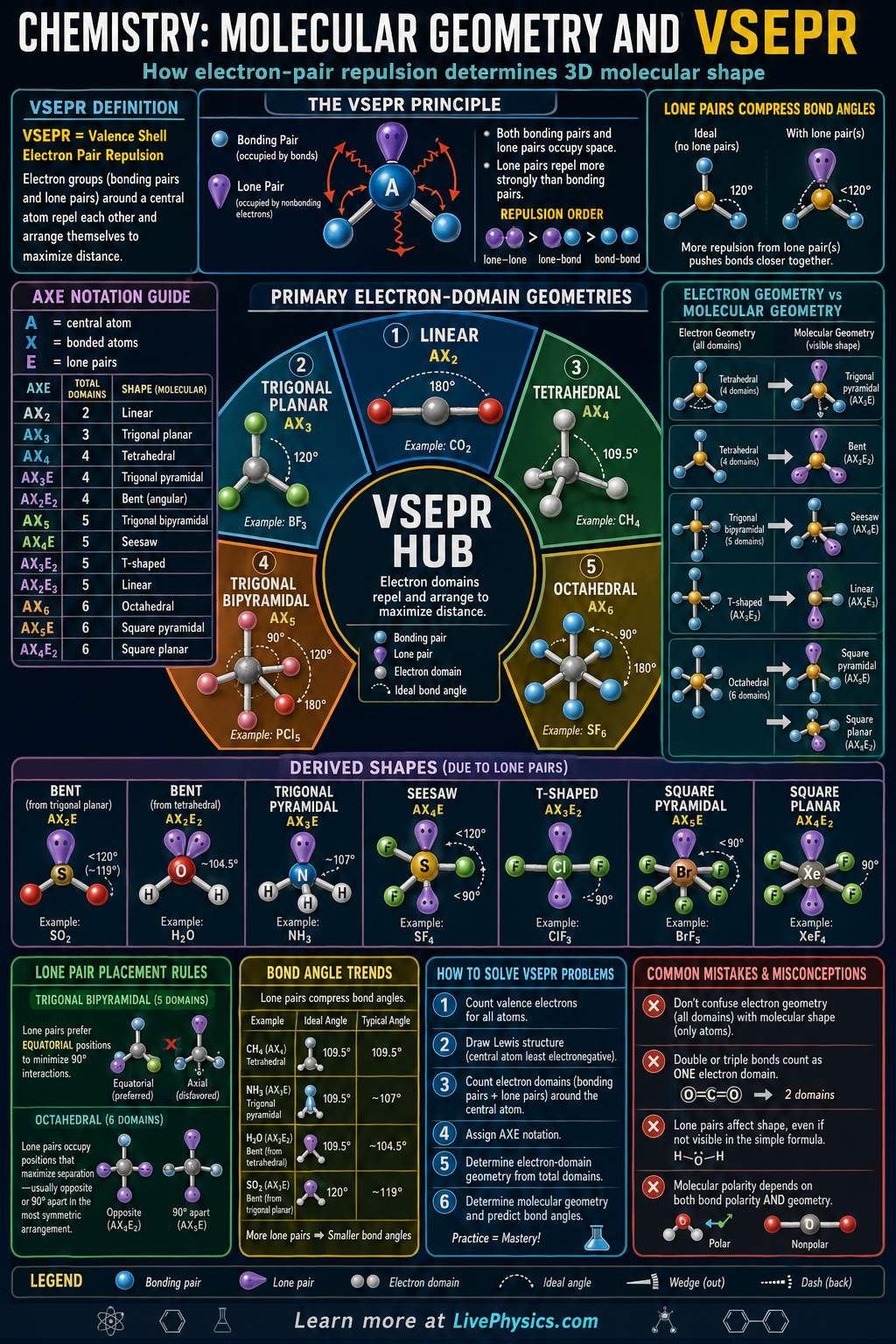
Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the three-dimensional shape of a molecule based on the principle that electron pairs - both bonding pairs and lone pairs - repel each other and arrange themselves as far apart as possible. The electron geometry describes the arrangement of all electron pairs around the central atom; the molecular geometry describes only the positions of the atoms (ignoring lone pairs).
Lone pairs exert more repulsion than bonding pairs, compressing bond angles below ideal values. Water (2 bonding pairs, 2 lone pairs) has tetrahedral electron geometry but bent molecular geometry with a 104.5° angle, less than the 109.5° ideal. Ammonia (3 bonding pairs, 1 lone pair) is trigonal pyramidal with 107° angles. Molecular geometry directly determines polarity: a symmetric molecule (like CO₂, linear) is nonpolar even if individual bonds are polar, because the dipoles cancel.
Key Facts
- VSEPR: electron pairs arrange to minimize repulsion; lone pairs repel more than bonding pairs
- 2 groups (0 lone pairs): linear, 180°
- 3 groups: trigonal planar (0 LP) 120°; bent (1 LP) ~117°
- 4 groups: tetrahedral (0 LP) 109.5°; trigonal pyramidal (1 LP) ~107°; bent (2 LP) ~104.5°
- 5 groups: trigonal bipyramidal; 6 groups: octahedral
- Polarity requires both polar bonds AND asymmetric geometry
Vocabulary
- VSEPR
- Valence Shell Electron Pair Repulsion theory; predicts molecular geometry by minimizing repulsion among electron pairs around the central atom.
- Electron geometry
- The three-dimensional arrangement of all electron pairs (bonding and lone) around the central atom.
- Molecular geometry
- The three-dimensional arrangement of atoms only (not lone pairs) around the central atom.
- Lone pair
- A pair of valence electrons on an atom that is not involved in a covalent bond; exerts greater repulsion than a bonding pair.
- Dipole moment
- A measure of the polarity of a bond or molecule; depends on charge separation and direction. Nonzero net dipole = polar molecule.
Common Mistakes to Avoid
- Confusing electron geometry with molecular geometry. If a central atom has 4 electron domains but 2 are lone pairs, the electron geometry is tetrahedral but the molecular geometry is bent.
- Forgetting to count lone pairs when applying VSEPR. Lone pairs are just as important as bonding pairs in determining the shape - the shape of water is not linear.
- Assuming a molecule with polar bonds is always polar. If the molecule is symmetric (e.g. BF₃ is trigonal planar, CO₂ is linear), the bond dipoles cancel and the molecule is nonpolar.
- Using bond order instead of electron domains for VSEPR. A double or triple bond counts as ONE electron domain (one bonding region), not two or three.
Practice Questions
- 1 Draw the Lewis structure of NH₃. Predict its electron geometry, molecular geometry, and approximate bond angles.
- 2 CO₂ has two polar C=O bonds. Explain why CO₂ is a nonpolar molecule using molecular geometry.
- 3 Predict the molecular geometry of SF₄ (sulfur with 4 F and 1 lone pair). What are the approximate F–S–F bond angles?