Chemical Bonding: Ionic and Covalent
Comparing how atoms bond and form compounds
Comparing how atoms bond and form compounds
Chemistry - Grade 9-12
- 1
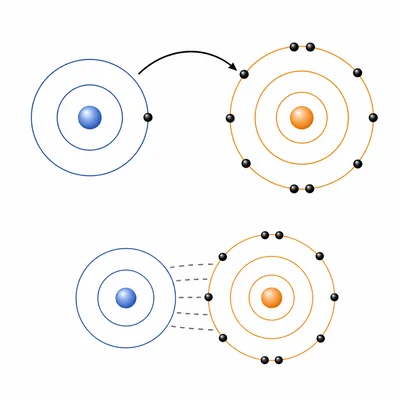
Explain how an ionic bond forms between atoms.
- 2
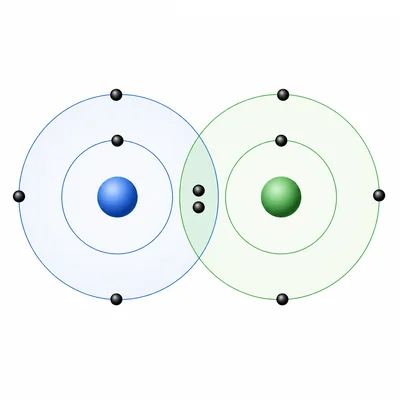
Explain how a covalent bond forms between atoms.
- 3
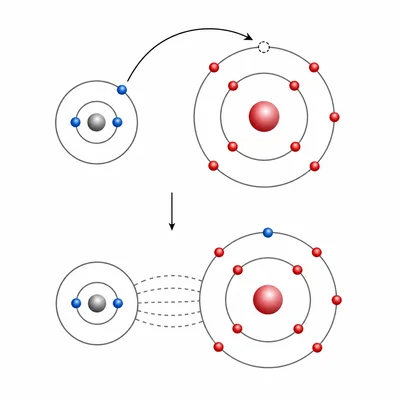
Classify the bond in sodium chloride, NaCl, as ionic or covalent and explain your choice.
- 4
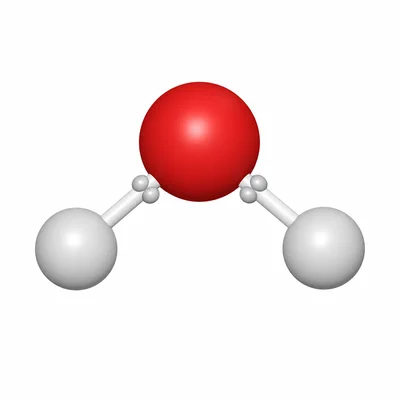
Classify the bond in water, H2O, as ionic or covalent and explain your choice.
- 5
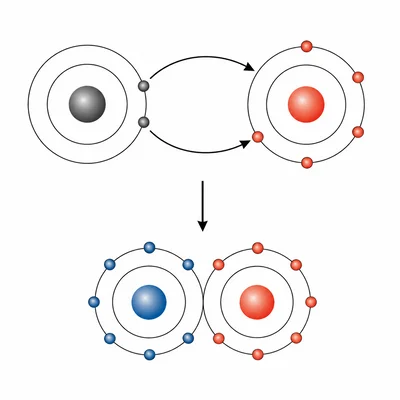
Magnesium reacts with oxygen to form magnesium oxide, MgO. Describe what happens to the electrons during bond formation.
- 6
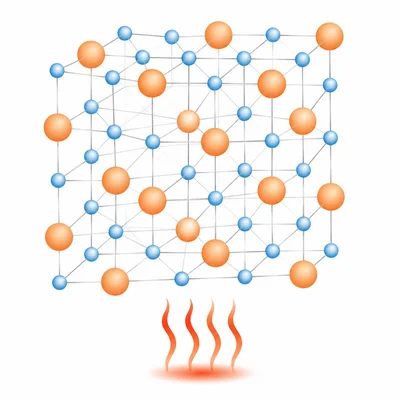
Why do ionic compounds usually have high melting points?
- 7
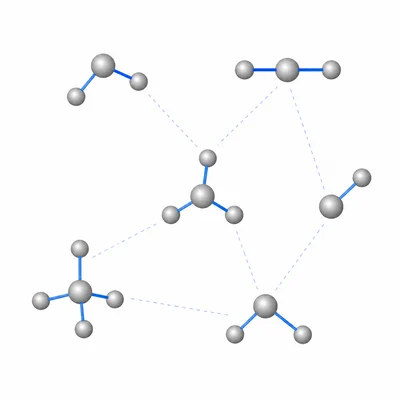
Why do many covalent compounds have lower melting and boiling points than ionic compounds?
- 8
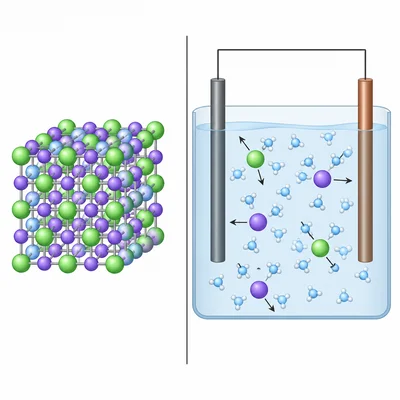
A compound conducts electricity when dissolved in water but not as a solid. Is it more likely ionic or covalent? Explain.
- 9
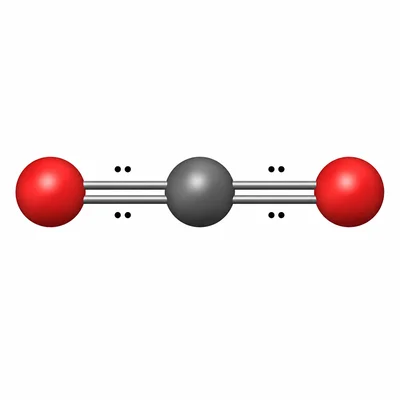
Carbon dioxide, CO2, is made of carbon and oxygen. Is this compound ionic or covalent? Explain how you know.
- 10
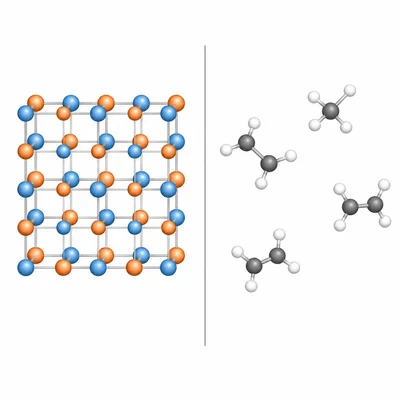
Compare the structure of an ionic compound with the structure of a molecular covalent compound.
- 11
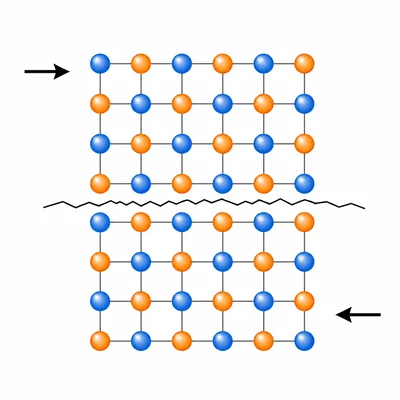
Predict whether potassium bromide, KBr, is likely to be brittle or flexible as a solid. Explain your answer.
- 12
Write one major difference and one similarity between ionic and covalent bonding.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems