Science: Atomic Structure and Electron Configuration
Understanding atoms, subatomic particles, and how electrons are arranged
Understanding atoms, subatomic particles, and how electrons are arranged
Chemistry - Grade 9-12
- 1
An atom of carbon has an atomic number of 6 and a mass number of 12. How many protons, neutrons, and electrons does a neutral carbon atom have?
- 2
An atom has 17 protons and 18 electrons. Identify the element and state whether the particle is a neutral atom or an ion.
- 3
Write the full electron configuration for a neutral oxygen atom.
- 4
Write the noble gas electron configuration for sodium.
- 5
How many valence electrons does a neutral sulfur atom have? Explain how you know from its electron configuration.
- 6
Write the full electron configuration for magnesium, which has 12 electrons.
- 7
A neutral atom has the electron configuration 1s2 2s2 2p6 3s2 3p5. Identify the element and state how many valence electrons it has.
- 8
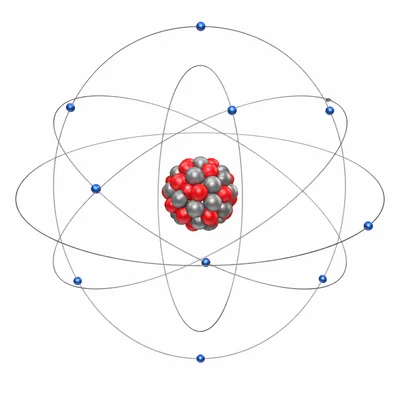
Compare the relative charges and locations of protons, neutrons, and electrons in an atom.
- 9
An aluminum atom has 13 protons and a mass number of 27. Find the number of neutrons. Then write the full electron configuration of a neutral aluminum atom.
- 10
Write the electron configuration for the fluoride ion, F-. Explain how it differs from the electron configuration of neutral fluorine.
- 11
Why are atoms electrically neutral when the number of protons equals the number of electrons?
- 12
A calcium atom is neutral and has 20 electrons. Write its noble gas electron configuration and state how many electrons are in its outermost energy level.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems