Science: Periodic Table Trends
Exploring patterns in atomic size, ionization energy, and electronegativity
Exploring patterns in atomic size, ionization energy, and electronegativity
Chemistry - Grade 9-12
- 1
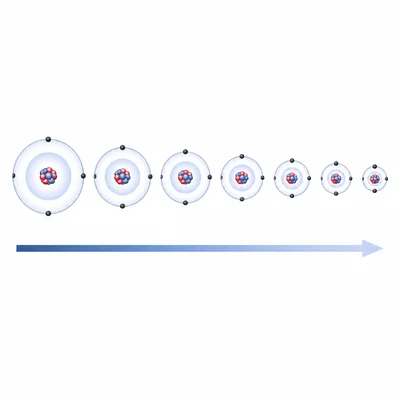
Describe the general trend in atomic radius as you move from left to right across a period.
- 2
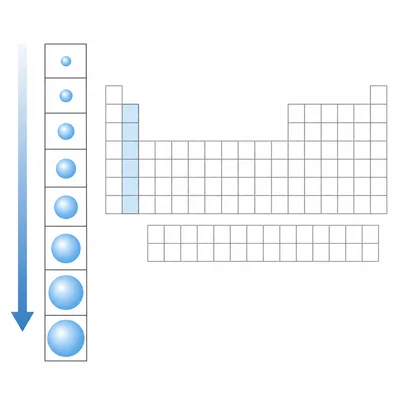
Describe the general trend in atomic radius as you move down a group.
- 3
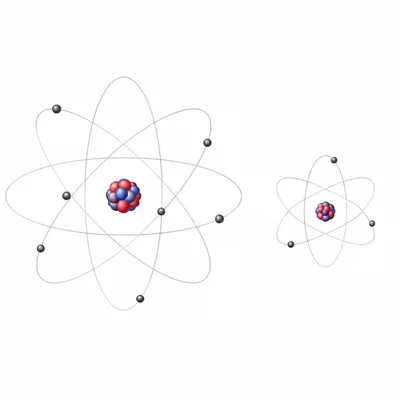
Which element has the larger atomic radius: sodium (Na) or chlorine (Cl)? Explain your answer.
- 4
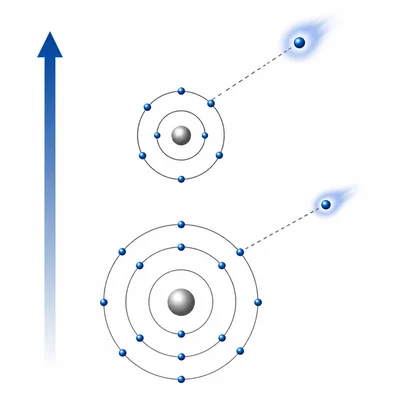
Which element has the higher first ionization energy: magnesium (Mg) or calcium (Ca)? Explain your answer.
- 5
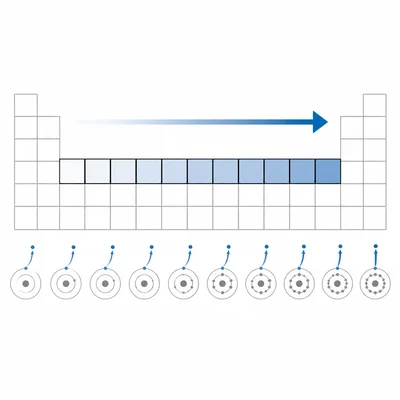
State the general trend in first ionization energy across a period from left to right.
- 6
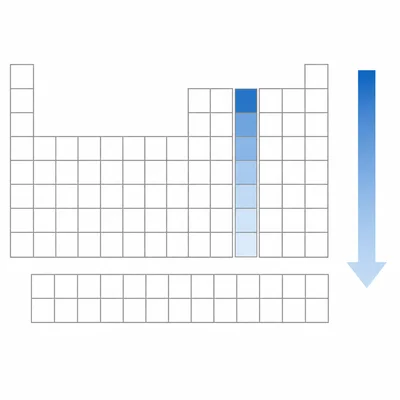
State the general trend in electronegativity as you move down a group.
- 7
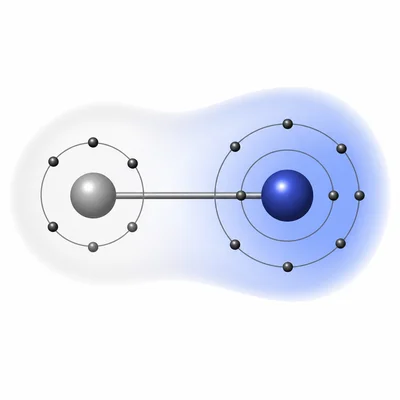
Which element is more electronegative: fluorine (F) or oxygen (O)? Explain your answer.
- 8
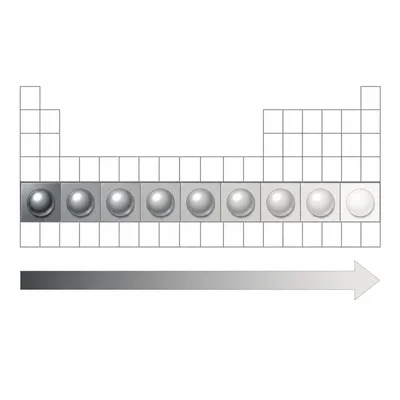
Compare the metallic character of elements as you move from left to right across a period.
- 9

Which element is more metallic: potassium (K) or bromine (Br)? Explain your answer.
- 10
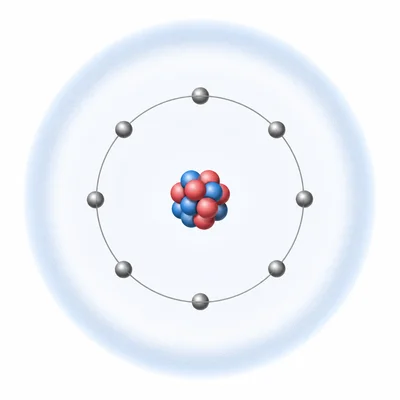
Explain why noble gases usually have very low chemical reactivity.
- 11
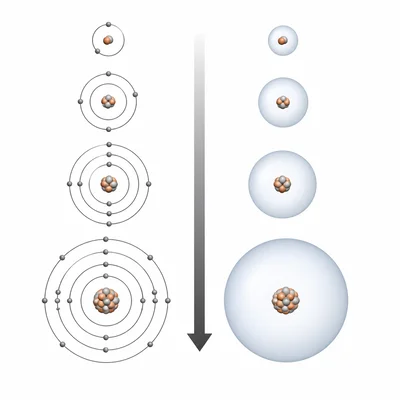
Which element would have the larger atomic radius: lithium (Li) or cesium (Cs)? Explain your answer.
- 12
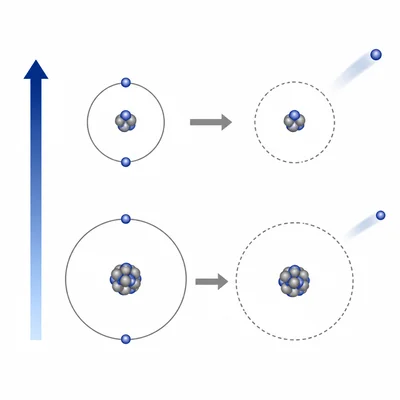
Which element has the higher ionization energy: neon (Ne) or argon (Ar)? Explain your answer.
- 13
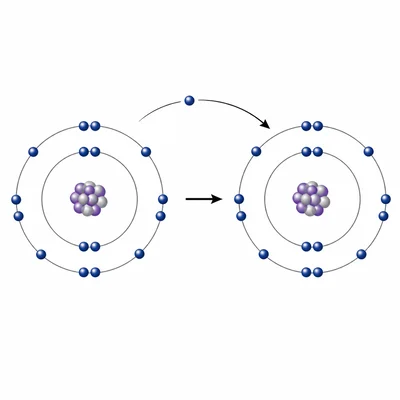
An element is located in Group 17 and Period 3. Predict whether it is likely to gain or lose an electron in a chemical reaction, and explain why.
- 14
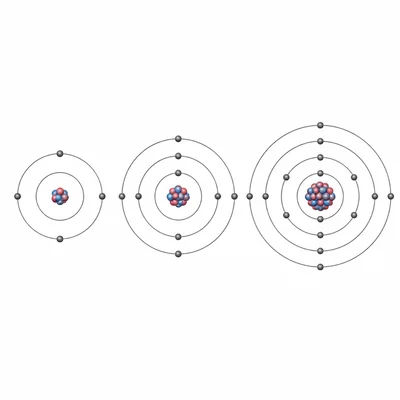
Why do elements in the same group often have similar chemical properties?
- 15
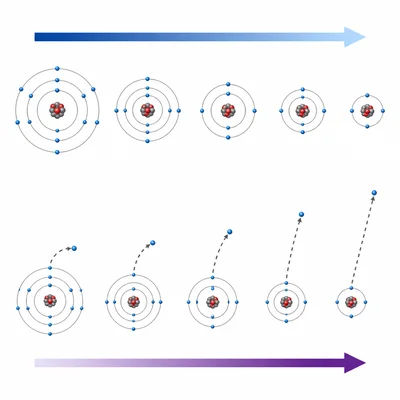
A student claims that atomic radius and ionization energy increase in the same direction across a period. Explain why this claim is incorrect.
Related Tools
Related Infographics
Related Mission Packs
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems