Chemistry: Covalent Bonding and Molecular Geometry (VSEPR)
Predicting molecular shapes from Lewis structures and electron domains
Predicting molecular shapes from Lewis structures and electron domains
Chemistry - Grade 9-12
- 1
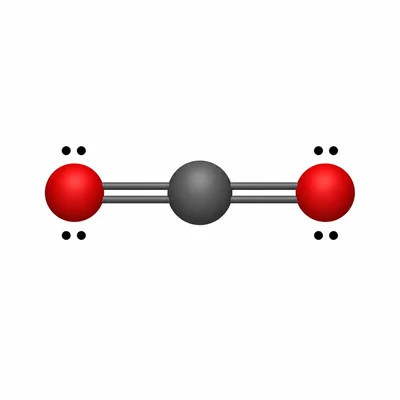
Draw the Lewis structure for CO2. Identify the electron-domain geometry, molecular geometry, approximate bond angle, and whether the molecule is polar or nonpolar.
- 2
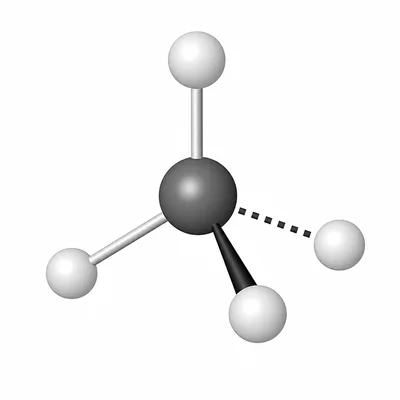
For CH4, count the electron domains around the central carbon atom and predict the molecular geometry and approximate H-C-H bond angle.
- 3
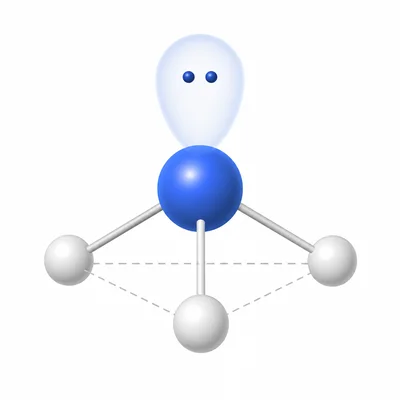
Explain why NH3 has a trigonal pyramidal molecular geometry instead of a trigonal planar geometry.
- 4
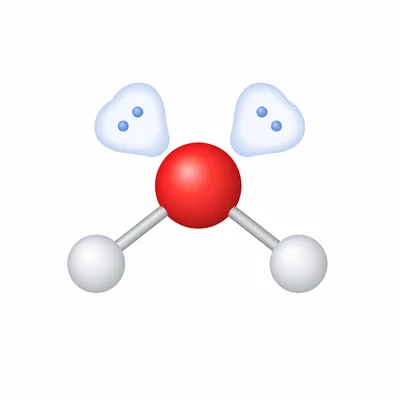
Water, H2O, has two O-H bonds and two lone pairs on oxygen. Predict its electron-domain geometry, molecular geometry, and approximate bond angle.
- 5
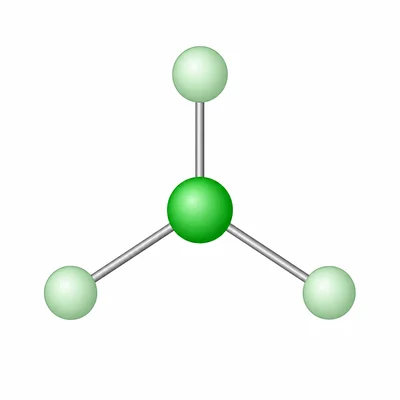
BF3 has boron as the central atom with three B-F bonds and no lone pairs on boron. Predict its molecular geometry, approximate bond angle, and polarity.
- 6
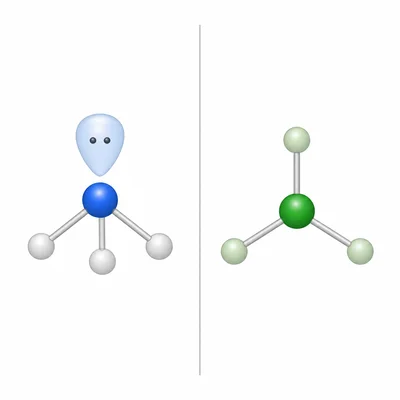
Compare NH3 and BF3. Both contain three atoms bonded to the central atom. Explain why they have different molecular geometries.
- 7
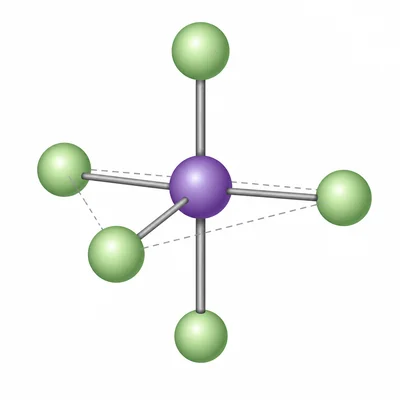
For PCl5, identify the number of electron domains around phosphorus, the molecular geometry, and the common bond angles.
- 8
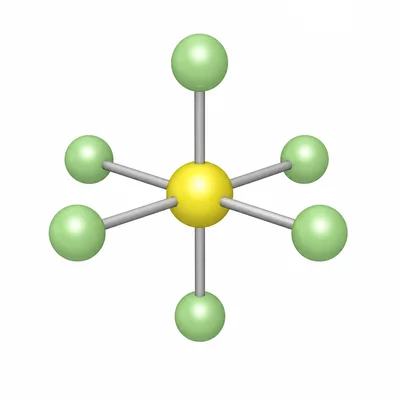
For SF6, identify the number of electron domains around sulfur, the molecular geometry, the common bond angles, and whether the molecule is polar or nonpolar.
- 9
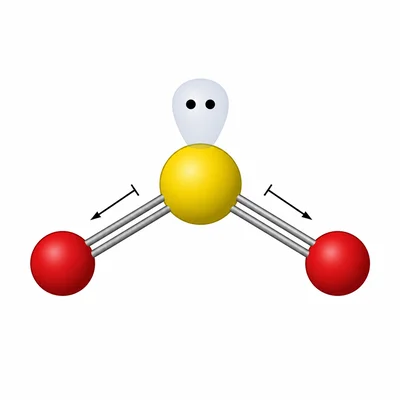
SO2 has sulfur as the central atom with two S-O bonding regions and one lone pair on sulfur. Predict the electron-domain geometry, molecular geometry, and whether SO2 is polar.
- 10
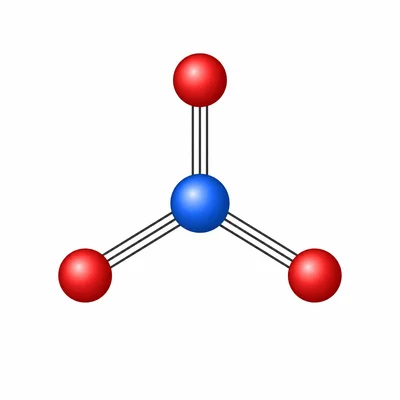
The nitrate ion, NO3-, has three equivalent N-O bonds around nitrogen and no lone pairs on nitrogen. Predict its molecular geometry and approximate bond angles.
- 11
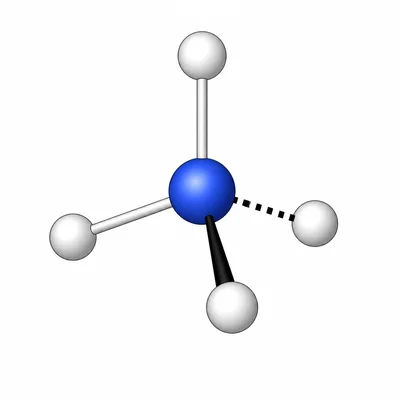
Draw the Lewis structure for NH4+. Then identify the molecular geometry and approximate H-N-H bond angle.
- 12
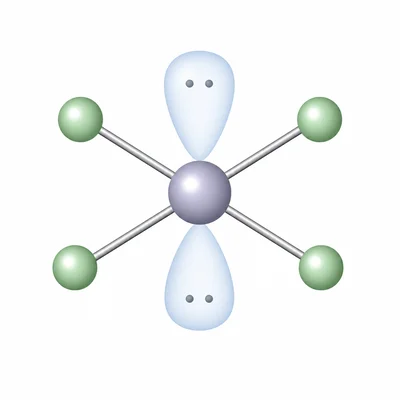
XeF4 has four Xe-F bonds and two lone pairs on xenon. Predict its electron-domain geometry, molecular geometry, and polarity.
- 13
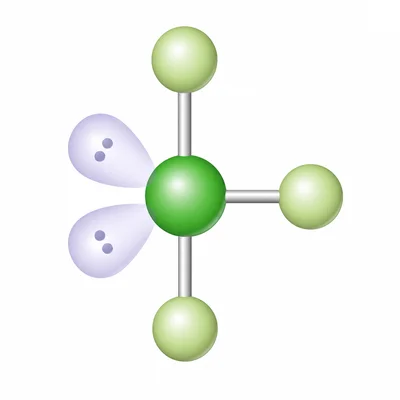
ClF3 has three Cl-F bonds and two lone pairs on chlorine. Use VSEPR to predict its molecular geometry.
- 14
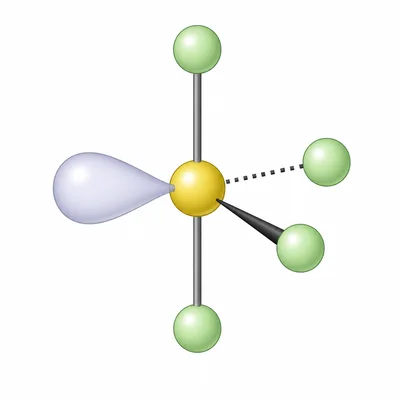
SF4 has four S-F bonds and one lone pair on sulfur. Predict its electron-domain geometry and molecular geometry.
- 15
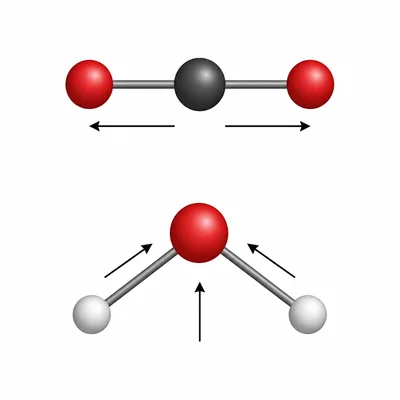
A student claims that every molecule with polar bonds must be a polar molecule. Use CO2 and H2O as examples to explain why this claim is not always true.
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems