States of Matter (Including Plasma)
Solid, Liquid, Gas, and Plasma: Particle Motion and Phase Changes
Related Tools
Related Labs
Related Worksheets
Related Cheat Sheets
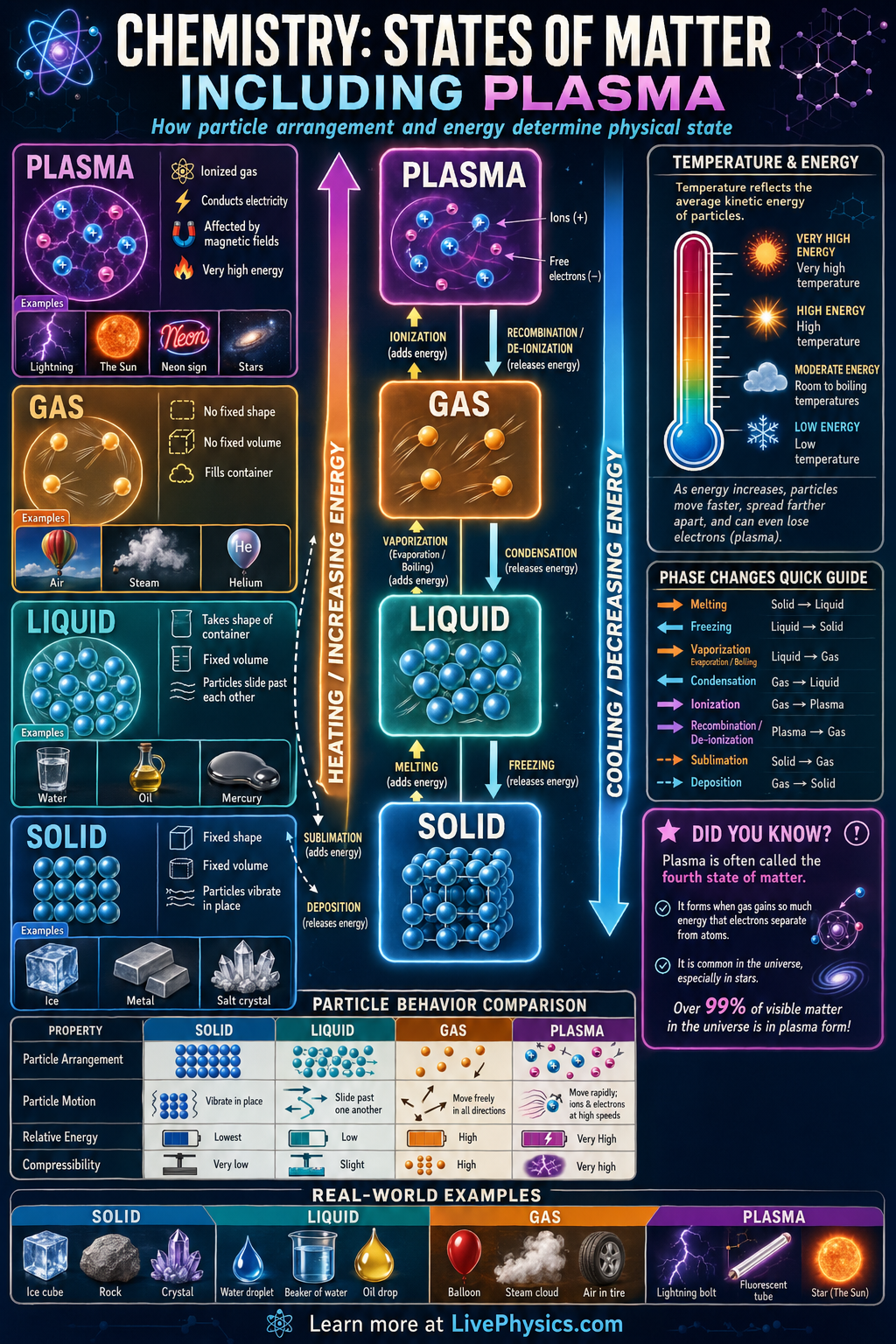
Matter exists in different states depending on how much energy its particles have and how strongly those particles attract one another. The four main states shown in basic chemistry are solid, liquid, gas, and plasma. Understanding these states helps explain everyday events such as melting ice, boiling water, and the glowing gas in lightning or neon signs. The state of a substance affects its shape, volume, density, and ability to flow.
As energy is added, particles move faster and can overcome some or all of the attractive forces holding them together. A solid can melt into a liquid, a liquid can vaporize into a gas, and a gas can become plasma if enough energy strips electrons from atoms. When energy is removed, the reverse changes can happen through condensation, freezing, and recombination. These changes are physical changes of state, not chemical changes in the identity of the substance.
Key Facts
- Energy increases from solid to liquid to gas to plasma.
- Solid: definite shape and definite volume.
- Liquid: no definite shape but definite volume.
- Gas: no definite shape and no definite volume.
- Plasma: ionized gas made of ions and free electrons.
Vocabulary
- Solid
- A state of matter with tightly packed particles that keeps a fixed shape and a fixed volume.
- Liquid
- A state of matter with particles that can slide past one another, giving it a fixed volume but no fixed shape.
- Gas
- A state of matter with widely spaced particles that move freely and fill the entire container.
- Plasma
- A high energy state of matter in which atoms are ionized into charged particles and free electrons.
- Phase change
- A physical change in which a substance moves from one state of matter to another without changing its chemical identity.
Common Mistakes to Avoid
- Thinking gas and plasma are the same, which is wrong because plasma contains charged particles created when electrons are stripped from atoms.
- Assuming melting, boiling, or freezing changes the substance into a new chemical, which is wrong because these are physical changes of state.
- Believing particles in a solid do not move at all, which is wrong because they still vibrate in place.
- Confusing heat with temperature, which is wrong because heat is energy transferred while temperature measures average kinetic energy of particles.
Practice Questions
- 1 A 200 g sample of ice absorbs energy and melts completely into liquid water without any mass loss. What is the mass of the liquid water after melting?
- 2 A gas sample has a mass of 12 g and occupies a volume of 6 cm. Using , what is its density?
- 3 Explain why plasma conducts electricity better than an ordinary neutral gas.