Chemistry: Reaction Mechanisms and Activation Energy
Connecting reaction pathways, rate-determining steps, and energy changes
Connecting reaction pathways, rate-determining steps, and energy changes
Chemistry - Grade 9-12
- 1
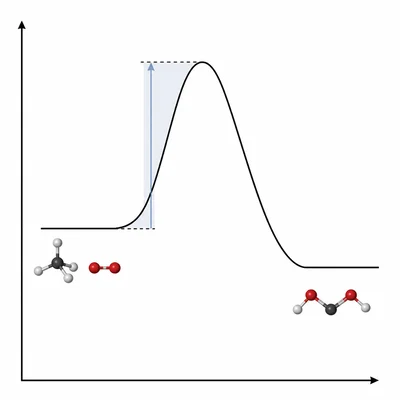
In your own words, explain what activation energy is and why it matters in a chemical reaction.
- 2
A reaction has the following mechanism: Step 1: A + B -> C, Step 2: C + D -> E. Write the overall reaction and identify any intermediate.
- 3
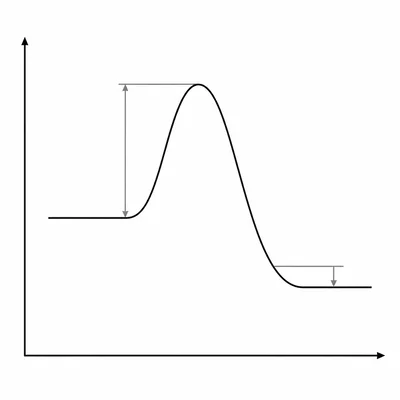
A potential energy diagram shows reactants at 40 kJ, a peak at 110 kJ, and products at 25 kJ. What is the activation energy for the forward reaction, and is the reaction exothermic or endothermic?
- 4
For the mechanism Step 1: NO2 + NO2 -> NO3 + NO, slow, Step 2: NO3 + CO -> NO2 + CO2, fast, write the overall reaction.
- 5
Using the same mechanism, Step 1: NO2 + NO2 -> NO3 + NO, slow, Step 2: NO3 + CO -> NO2 + CO2, fast, what rate law is predicted by the slow elementary step?
- 6
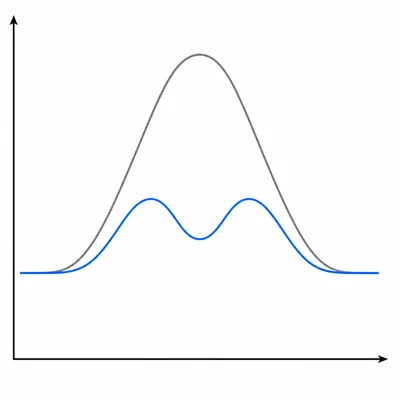
A catalyst is added to a reaction mixture. Explain how the catalyst changes the reaction mechanism and activation energy.
- 7
In a reaction mechanism, how can you tell the difference between a catalyst and an intermediate?
- 8
A reaction has two steps. Step 1 has an activation energy of 35 kJ and Step 2 has an activation energy of 80 kJ. Which step is most likely the rate-determining step? Explain.
- 9
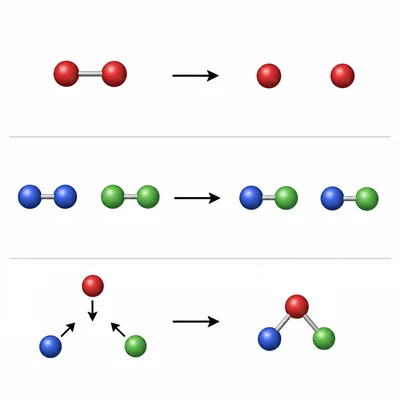
Classify each elementary step by molecularity: Step 1: A -> products, Step 2: A + B -> products, Step 3: 2A + B -> products.
- 10
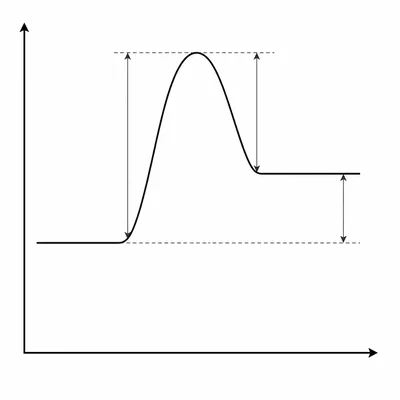
A reaction has reactants at 60 kJ, products at 100 kJ, and a transition state at 150 kJ. Calculate the forward activation energy, reverse activation energy, and enthalpy change.
- 11
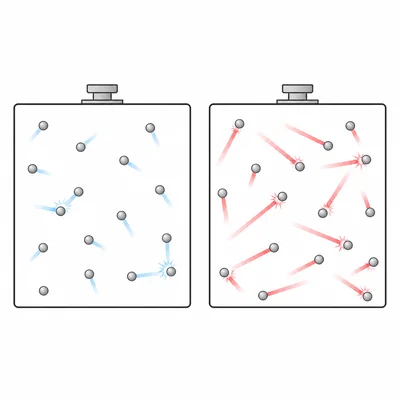
Explain why increasing temperature usually increases the rate of a chemical reaction.
- 12
For the elementary reaction H2 + I2 -> 2HI, write the rate law for this single-step process.
- 13
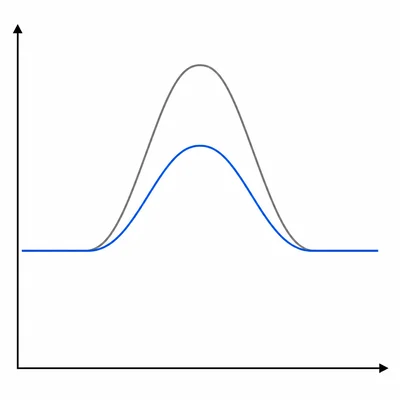
A student says, "A catalyst increases the amount of product formed at equilibrium because it lowers activation energy." Explain what is correct and what is incorrect in this statement.
- 14
Consider the mechanism Step 1: X + Y -> Z, fast, Step 2: Z + Y -> W, slow. Why might the experimentally measured rate law not be simply rate = k[Z][Y]?
- 15
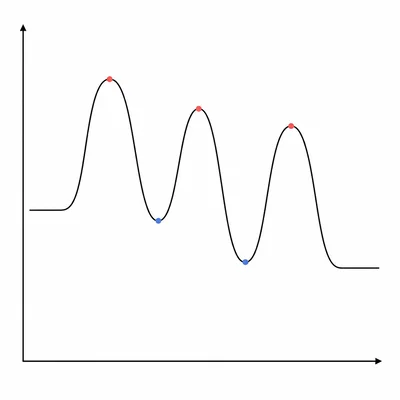
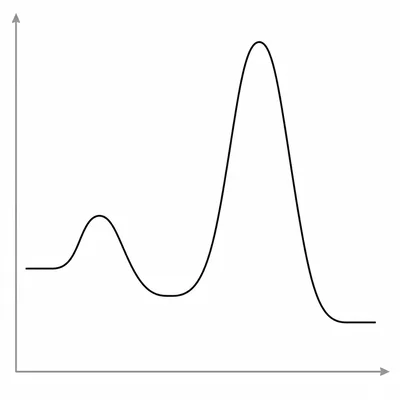
A reaction coordinate diagram has three peaks and two valleys between the reactants and products. What do the peaks and valleys represent in a multi-step mechanism?
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems