Physics: Photoelectric Effect and Wave-Particle Duality
Connecting photons, electrons, and matter waves
Connecting photons, electrons, and matter waves
Physics - Grade 9-12
- 1
A beam of green light has a wavelength of 500 nm. Calculate the energy of one photon in joules and in electron volts.
- 2
A metal has a threshold frequency of 5.00 x 10^14 Hz. Calculate its work function in joules and in electron volts.
- 3
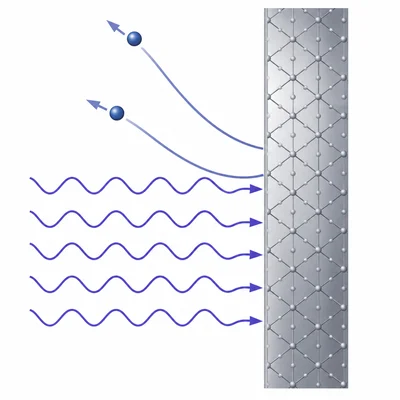
Light with a wavelength of 400 nm shines on a metal with a work function of 2.0 eV. Determine whether electrons are emitted, and find the maximum kinetic energy of the emitted electrons.
- 4
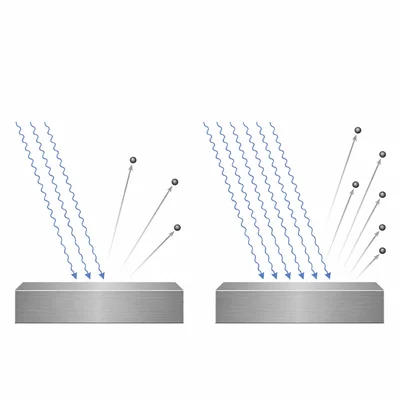
A metal is illuminated by light with a frequency above its threshold frequency. The intensity of the light is doubled while the frequency stays the same. Explain what happens to the number of emitted electrons and to their maximum kinetic energy.
- 5
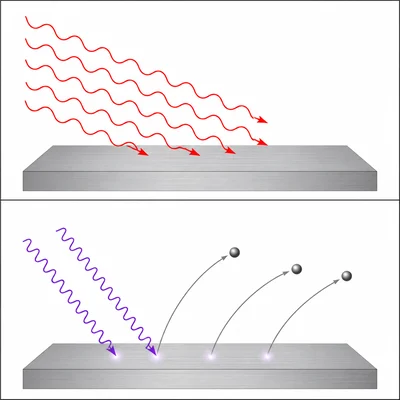
Bright red light does not eject electrons from a metal surface, but dim violet light does. Explain why this supports the photon model of light.
- 6
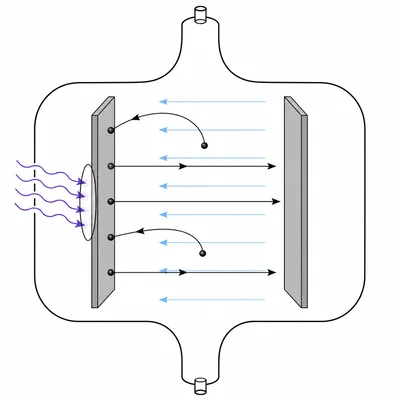
Photoelectrons from a metal have a maximum kinetic energy of 0.85 eV. What stopping potential is needed to stop the most energetic electrons?
- 7
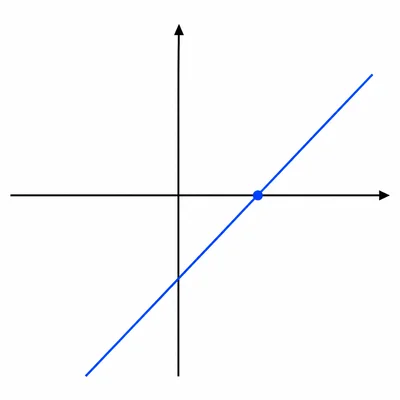
A graph of maximum kinetic energy of photoelectrons versus light frequency is a straight line. State what the slope and the x-intercept represent.
- 8
An electron with mass 9.11 x 10^-31 kg moves at 2.00 x 10^6 m/s. Calculate its de Broglie wavelength.
- 9
A 0.145 kg baseball travels at 40.0 m/s. Calculate its de Broglie wavelength and explain why its wave behavior is not noticeable in everyday life.
- 10
Which has greater photon energy: a microwave photon with frequency 1.00 x 10^10 Hz or a visible light photon with frequency 6.00 x 10^14 Hz? Explain your answer.
- 11
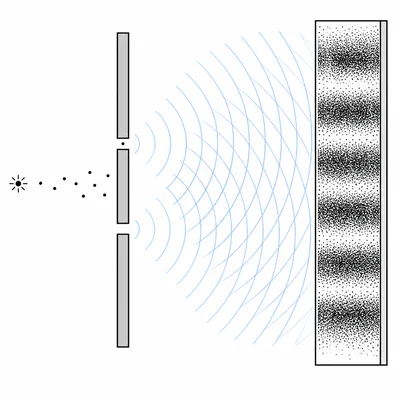
In a double-slit experiment, very dim light is sent through the slits so that photons arrive one at a time. Over time, an interference pattern appears on the screen. Explain how this shows wave-particle duality.
- 12
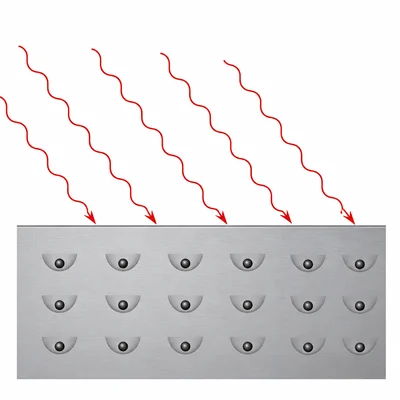
Classical wave theory predicted that a very bright low-frequency light beam should eventually eject electrons from a metal. In the photoelectric effect, this does not happen if the frequency is below the threshold frequency. Explain why.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Physics Worksheets
Forces & Motion
Grade 6-8 · 8 problems
Science: Energy Thermodynamics and Heat Transfer
Grade 9-12 · 15 problems
Science: Light and Sound
Grade 4-5 · 15 problems
Science: Newtons Laws of Motion
Grade 9-12 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems