Science: Nuclear Physics and Radioactivity
Atomic nuclei, decay, half-life, and radiation safety
Atomic nuclei, decay, half-life, and radiation safety
Physics - Grade 9-12
- 1
Define the term isotope and explain how isotopes of the same element are different from one another.
- 2
Carbon-14 has 6 protons and 8 neutrons. Write its mass number and explain what the number means.
- 3
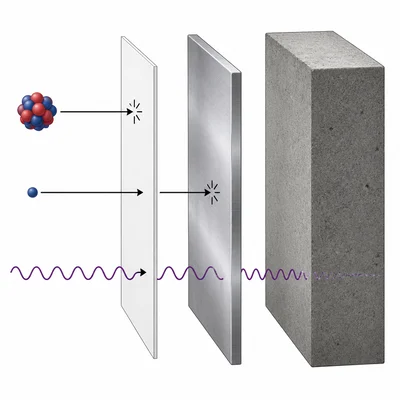
Compare alpha, beta, and gamma radiation by describing what each one is and how penetrating it is.
- 4
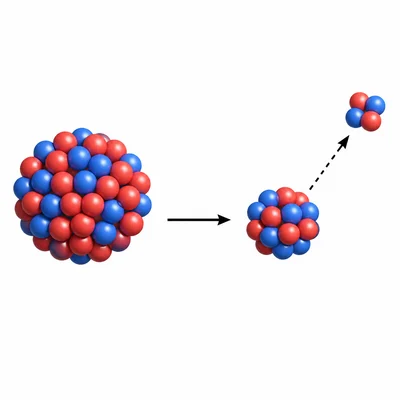
During alpha decay, what happens to the atomic number and mass number of the nucleus?
- 5
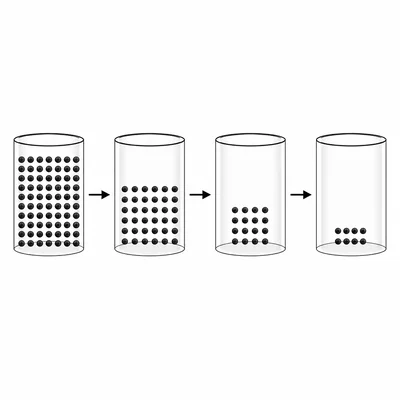
A radioactive sample has a half-life of 10 years. If the sample starts with 80 grams, how much will remain after 30 years?
- 6
Explain why radioactive decay is considered a random process for a single nucleus but predictable for a large sample.
- 7
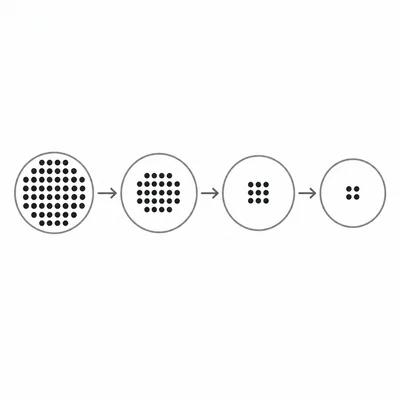
If a radioisotope has a half-life of 6 hours, what fraction of the original sample remains after 18 hours? Also state the percent remaining.
- 8
Describe one medical use of radioactivity and explain why it is useful in that application.
- 9
State the law of conservation of charge and mass number as it applies to nuclear equations.
- 10
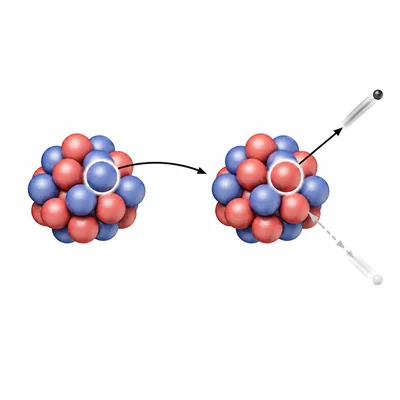
A nucleus emits beta-minus radiation. Describe what happens inside the nucleus during this process.
- 11
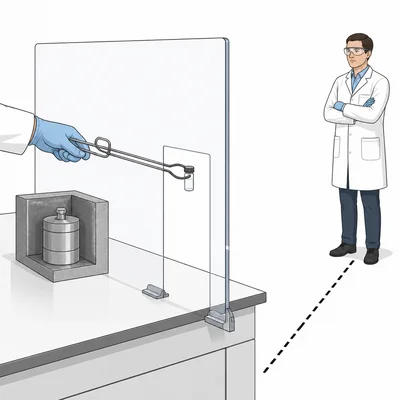
List two safety practices used when working with radioactive materials and explain how each one reduces exposure.
- 12
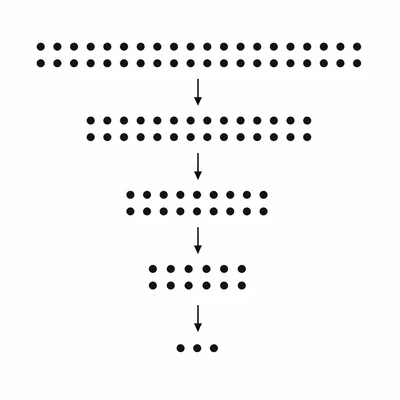
A sample begins with 1600 undecayed nuclei. After 4 half-lives, how many undecayed nuclei remain?
Related Infographics
Related Cheat Sheets
More Physics Worksheets
Forces & Motion
Grade 6-8 · 8 problems
Science: Energy Thermodynamics and Heat Transfer
Grade 9-12 · 15 problems
Science: Light and Sound
Grade 4-5 · 15 problems
Science: Newtons Laws of Motion
Grade 9-12 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems