Nuclear Physics & Radioactivity Cheat Sheet
A printable reference covering nuclear notation, radioactive decay, half-life, activity, binding energy, mass defect, and nuclear reactions for grades 11-12.
Related Tools
Related Worksheets
Related Infographics
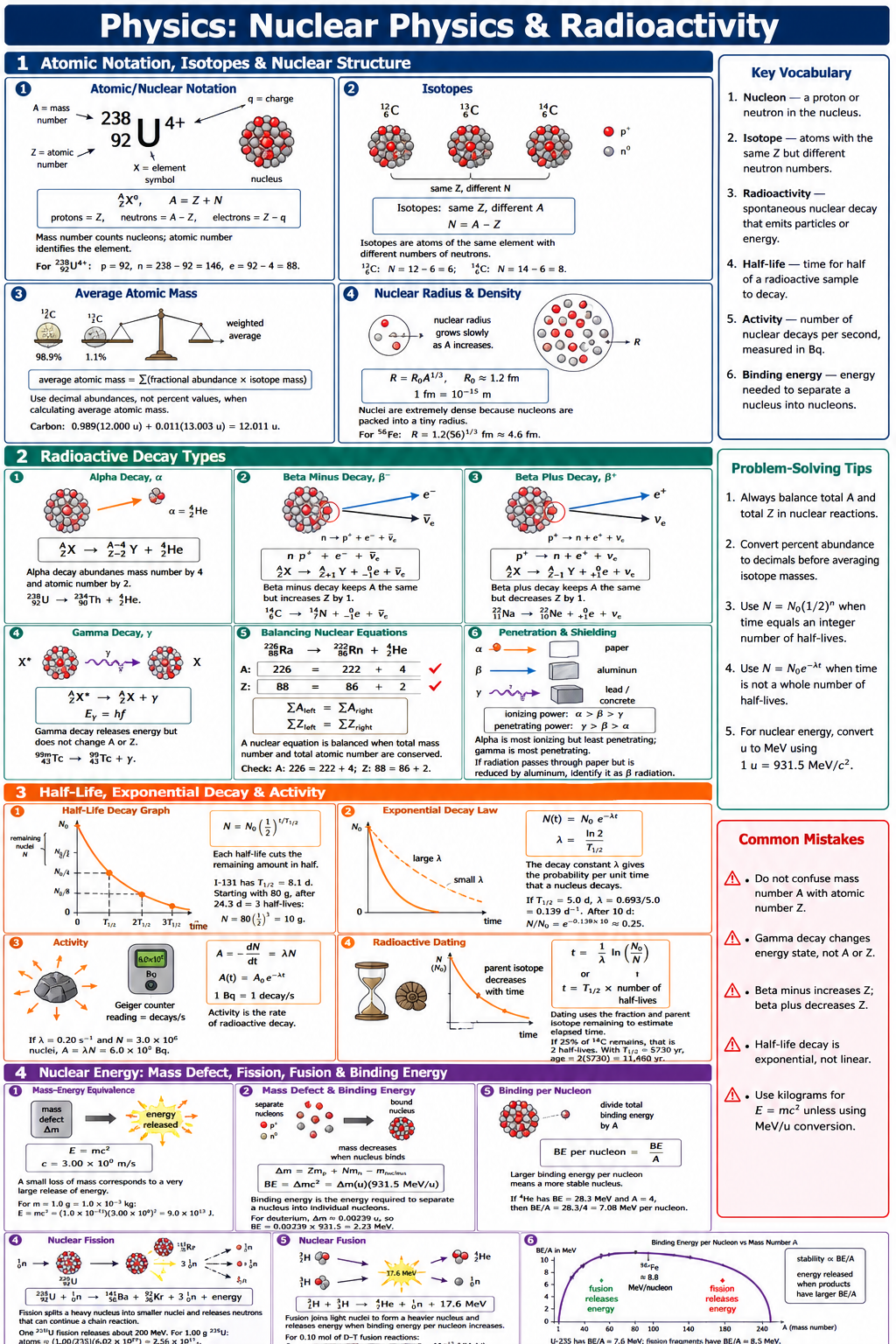
Nuclear physics studies the structure, stability, and transformations of atomic nuclei. This cheat sheet helps students track nuclear symbols, balance decay equations, and connect radioactive decay to measurable quantities. It is especially useful for solving half-life, activity, binding energy, and reaction energy problems in grade 11 and 12 physics. The most important ideas are conservation of nucleon number, conservation of charge, and exponential decay. Radioactive samples follow , with half-life given by . Nuclear energy calculations use mass defect and Einstein’s relation , often expressed using .
Key Facts
- A nuclide is written as , where is the mass number, is the atomic number, and the number of neutrons is .
- In every nuclear reaction, total mass number and total atomic number are conserved, so and .
- Alpha decay emits , so a parent nucleus changes according to .
- Beta-minus decay emits an electron and an antineutrino, so .
- Beta-plus decay emits a positron and a neutrino, so .
- Radioactive decay follows , where is the number of undecayed nuclei, is the initial number, and is the decay constant.
- Half-life and decay constant are related by , and after half-lives the remaining amount is .
- Nuclear binding energy is found from , where the mass defect is .
Vocabulary
- Nuclide
- A specific nucleus identified by its number of protons and total nucleons .
- Isotope
- Atoms of the same element with the same but different neutron numbers .
- Half-life
- The time required for half of the radioactive nuclei in a sample to decay.
- Activity
- The decay rate of a radioactive sample, given by .
- Mass defect
- The missing mass between separate nucleons and the actual nucleus, converted into binding energy.
- Binding energy
- The energy required to completely separate a nucleus into individual protons and neutrons.
Common Mistakes to Avoid
- Forgetting to conserve both and is wrong because nuclear equations must balance nucleon number and charge separately.
- Confusing half-life with decay constant is wrong because is a time while is a probability per unit time, related by .
- Using linear subtraction for radioactive decay is wrong because radioactive decay is exponential, so the correct model is or .
- Treating gamma emission as a change in atomic number is wrong because gamma decay releases energy but leaves and unchanged.
- Using atomic masses without checking electrons is wrong because mass defect calculations require consistent masses, either all nuclear masses or correctly balanced atomic masses.
Practice Questions
- 1 Complete the alpha decay equation: .
- 2 A sample has nuclei and a half-life of . How many nuclei remain after ?
- 3 A radioactive isotope has and nuclei. Find its activity using .
- 4 Explain why nuclei with higher binding energy per nucleon are generally more stable than nuclei with lower binding energy per nucleon.