Organic Chemistry Functional Groups
Organic Chemistry Functional Groups
Related Tools
Related Worksheets
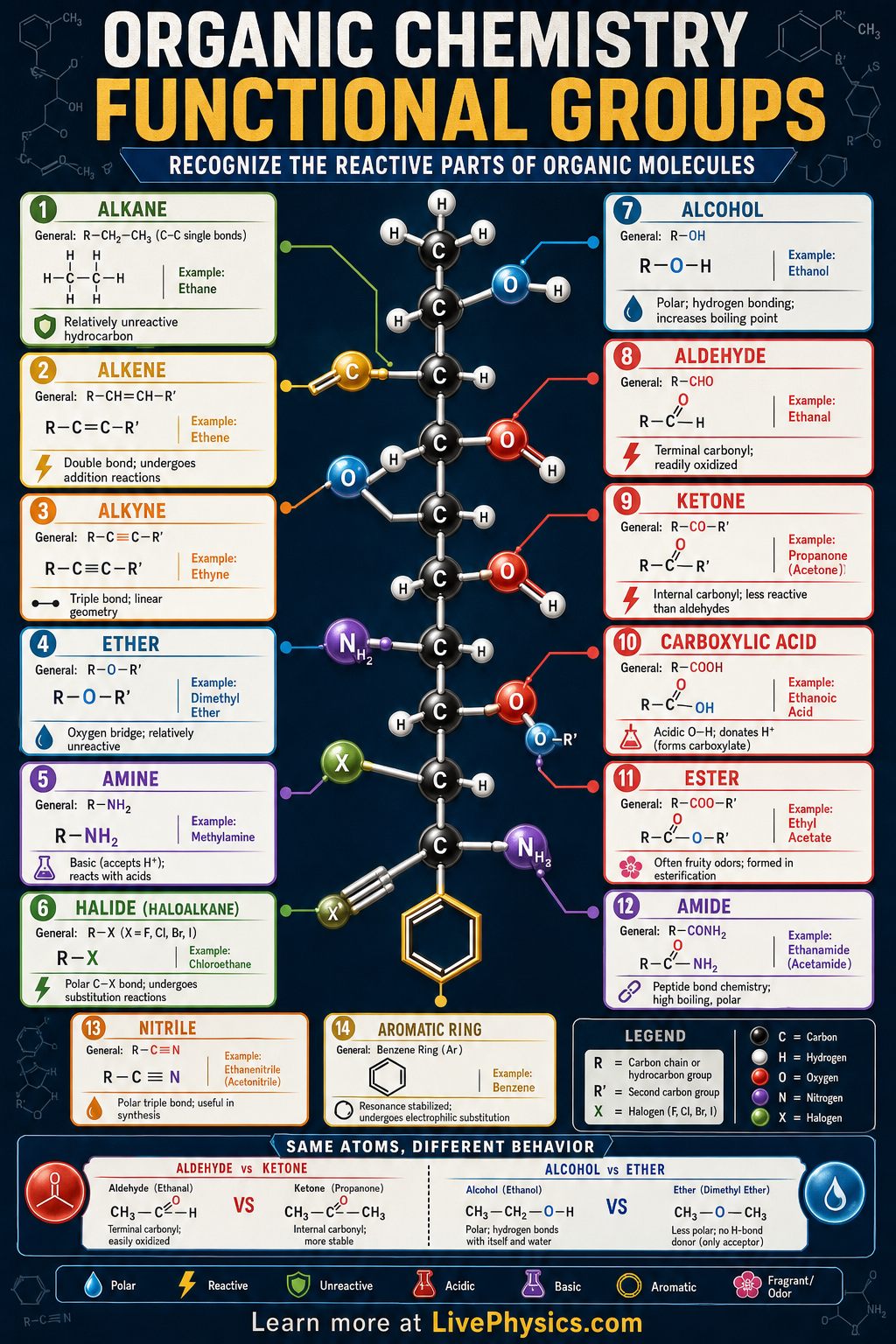
Functional groups are the reactive parts of organic molecules that give compounds their chemical behavior. A long carbon chain can be fairly unreactive, but adding an -OH, -COOH, -NH2, or C=O group can strongly change its boiling point, solubility, acidity, and reactions. Learning functional groups helps students recognize patterns instead of memorizing every organic molecule one by one.
Each functional group contains specific atoms and bonding patterns that control electron distribution. Polar bonds, lone pairs, and multiple bonds create sites where molecules can attract protons, donate electrons, or react with other compounds. In biology, medicine, fuels, plastics, and food chemistry, functional groups explain why molecules dissolve, smell, taste, polymerize, or interact with enzymes.
Key Facts
- Alcohols contain a hydroxyl group: R-OH.
- Carboxylic acids contain a carboxyl group: R-COOH, and can dissociate as RCOOH ⇌ RCOO- + H+.
- Aldehydes and ketones both contain a carbonyl group: C=O.
- Esters have the general structure R-COOR' and often form from a carboxylic acid plus an alcohol.
- Amines contain nitrogen bonded to carbon and hydrogen: R-NH2, R2NH, or R3N.
- Alkenes contain C=C and alkynes contain C≡C, making them more reactive than alkanes.
Vocabulary
- Functional group
- A specific atom or group of atoms in an organic molecule that largely determines its chemical reactions and properties.
- Hydroxyl group
- An -OH group covalently bonded to a carbon atom, commonly found in alcohols.
- Carbonyl group
- A C=O group found in aldehydes, ketones, carboxylic acids, esters, amides, and related compounds.
- Carboxyl group
- A -COOH group that combines a carbonyl and hydroxyl on the same carbon and usually behaves as a weak acid.
- Amine
- An organic compound with nitrogen bonded to carbon atoms, often acting as a weak base.
Common Mistakes to Avoid
- Calling every -OH group a hydroxide ion: an alcohol has -OH covalently bonded inside a neutral molecule, while hydroxide is the separate ion OH-.
- Confusing aldehydes and ketones: an aldehyde has the carbonyl carbon at the end of a carbon chain, while a ketone has the carbonyl carbon within the chain.
- Labeling every C=O as a carboxylic acid: a carboxylic acid must have both C=O and -OH attached to the same carbon, making -COOH.
- Ignoring the R group notation: R is not an element, but a placeholder for the rest of the carbon-containing molecule.
Practice Questions
- 1 1-propanol has the formula C3H8O. Using C = 12.01 g/mol, H = 1.008 g/mol, and O = 16.00 g/mol, calculate its molar mass.
- 2 Acetic acid, CH3COOH, reacts with ethanol to form an ester and water. If 0.50 mol of acetic acid produces 0.50 mol of water, how many grams of water form? Use H2O = 18.02 g/mol.
- 3 A molecule contains both a C=C double bond and a -COOH group. Explain how each functional group could affect the molecule's reactions or physical properties.