Chemistry: Organic Chemistry: Nomenclature and Isomers
Practice naming organic compounds and identifying isomers
Practice naming organic compounds and identifying isomers
Chemistry - Grade 9-12
- 1
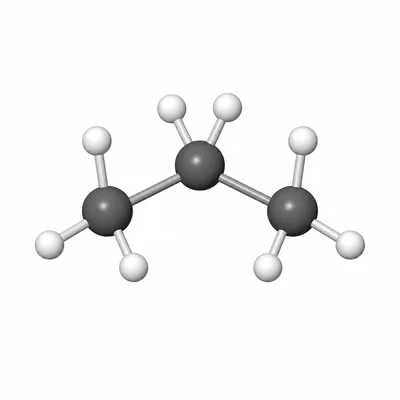
Name the compound with the condensed structural formula CH3-CH2-CH3.
- 2
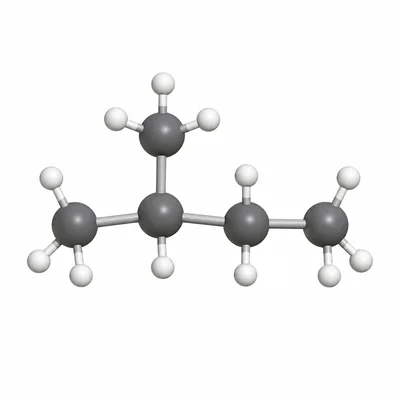
Name the compound with the condensed structural formula CH3-CH(CH3)-CH2-CH3.
- 3
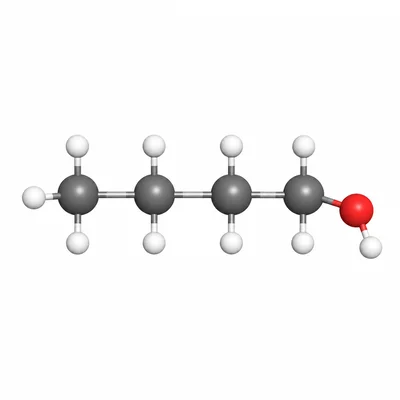
Name the alcohol with the condensed structural formula CH3-CH2-CH2-CH2-OH.
- 4
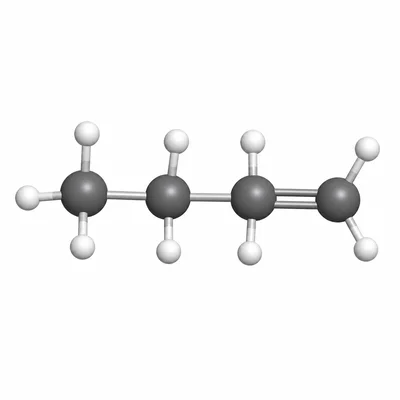
Name the alkene with the condensed structural formula CH3-CH2-CH=CH2.
- 5
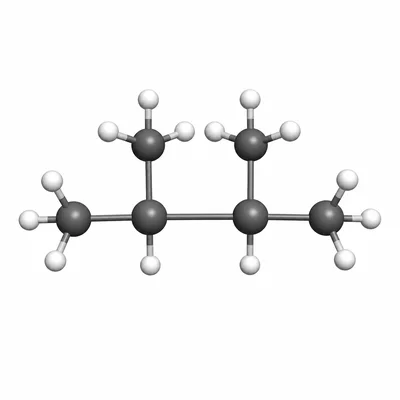
Name the compound with the condensed structural formula CH3-CH(CH3)-CH(CH3)-CH3.
- 6
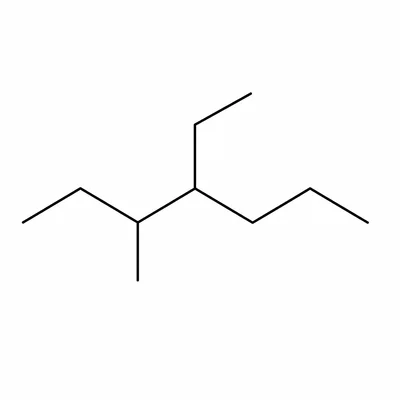
Write a condensed structural formula for 3-ethyl-2-methylhexane.
- 7
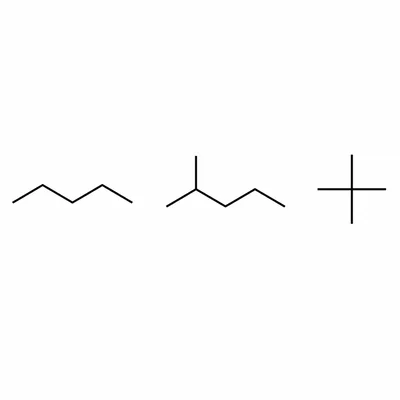
List all three structural isomers of C5H12 by IUPAC name.
- 8
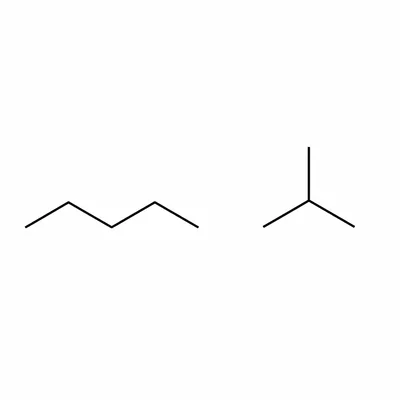
Explain what structural isomers are, using C4H10 as an example.
- 9
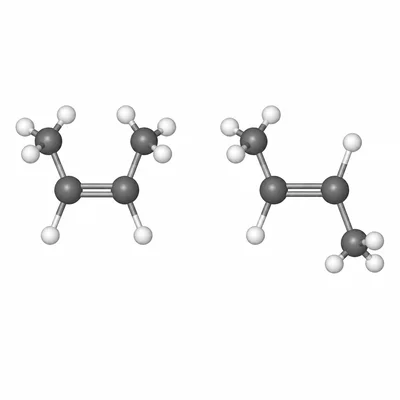
Describe the difference between cis-2-butene and trans-2-butene.
- 10
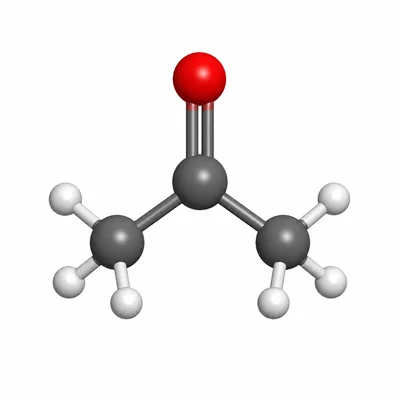
Name the compound CH3-CO-CH3 and identify its functional group.
- 11
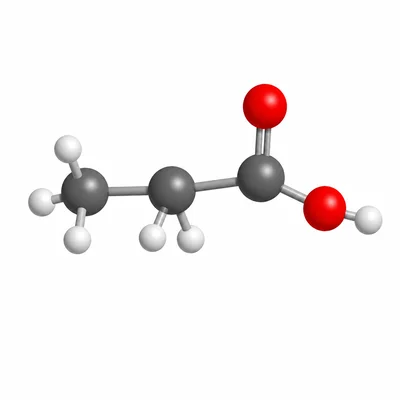
Name the compound CH3-CH2-COOH and identify its functional group.
- 12
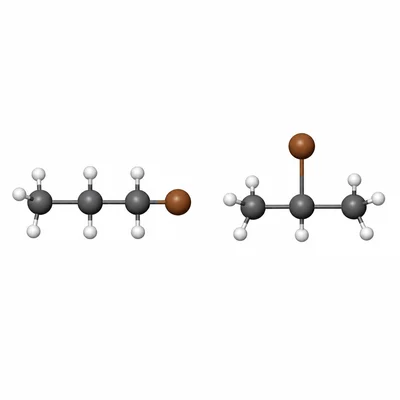
Name CH3-CH2-CH2-Br and give the name of one structural isomer with the same molecular formula.
- 13
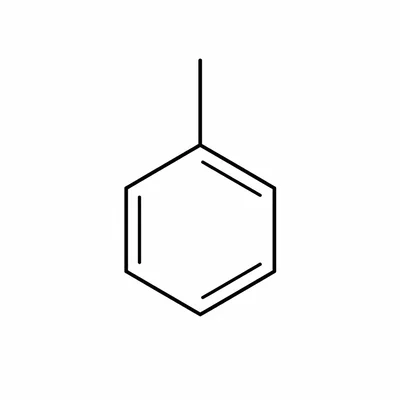
Name a benzene ring with one methyl group attached.
- 14
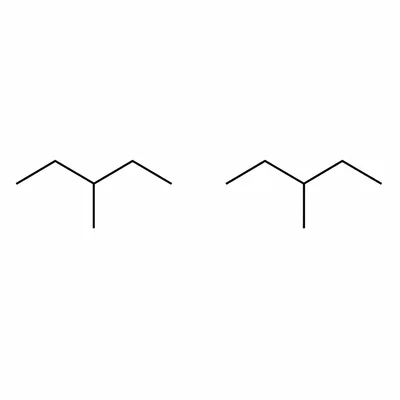
Are 2-methylpentane and 3-methylpentane structural isomers? Explain your answer.
- 15
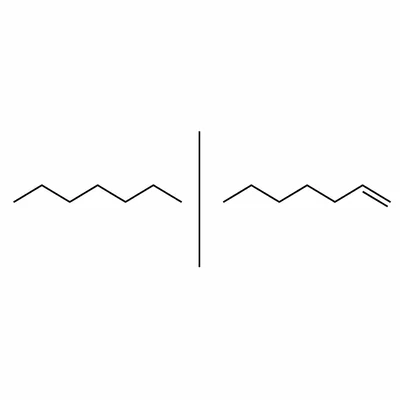
Explain the difference between a saturated hydrocarbon and an unsaturated hydrocarbon. Use hexane and hex-1-ene as examples.
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems