Chemistry: Intermolecular Forces and Physical Properties
Connecting particle attractions to boiling point, solubility, vapor pressure, and more
Connecting particle attractions to boiling point, solubility, vapor pressure, and more
Chemistry - Grade 9-12
- 1
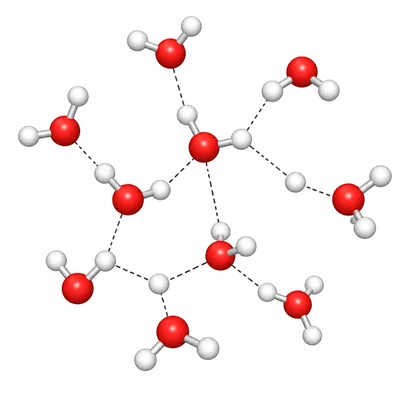
Identify the strongest intermolecular force present in a sample of liquid water, H2O, and explain why it occurs.
- 2
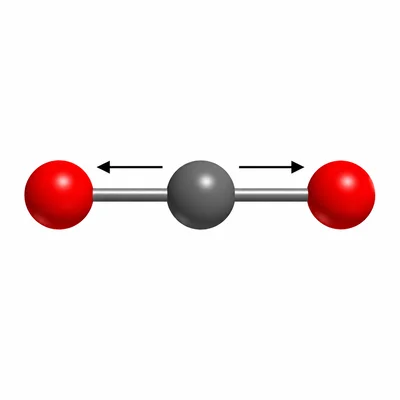
Identify the strongest intermolecular force present between molecules of carbon dioxide, CO2. Explain why CO2 does not have dipole-dipole attractions even though each C=O bond is polar.
- 3
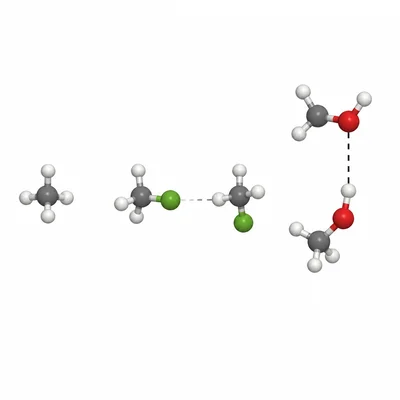
Rank the following substances from lowest boiling point to highest boiling point: CH4, CH3Cl, CH3OH. Explain your ranking using intermolecular forces.
- 4
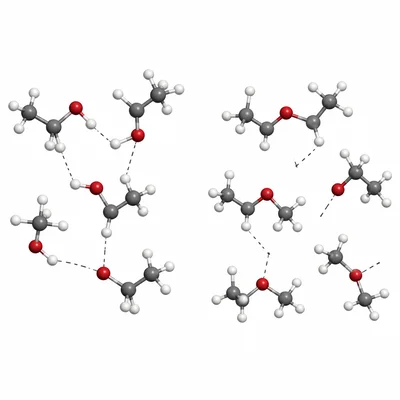
Explain why ethanol, C2H5OH, has a higher boiling point than dimethyl ether, CH3OCH3, even though they have the same molecular formula, C2H6O.
- 5
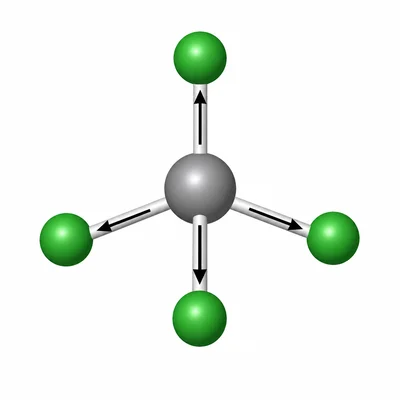
A student says, "All molecules with polar bonds are polar molecules." Explain why this statement is not always correct, using CCl4 as an example.
- 6
Compare the boiling points of F2 and Br2. Both are nonpolar diatomic molecules. Which one has the higher boiling point, and why?
- 7
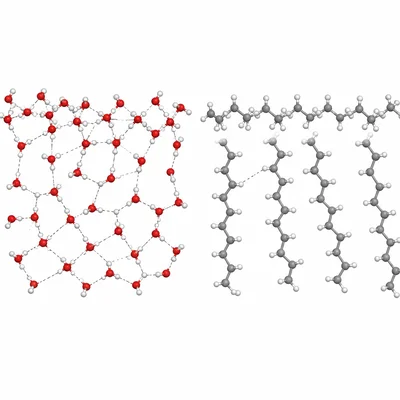
Explain why water has a much higher surface tension than hexane, C6H14.
- 8
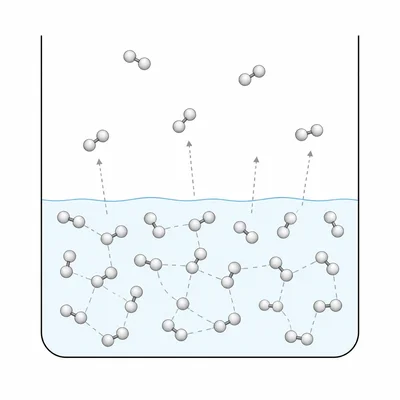
A liquid has a high vapor pressure at room temperature. What does this tell you about the strength of its intermolecular forces? Explain.
- 9
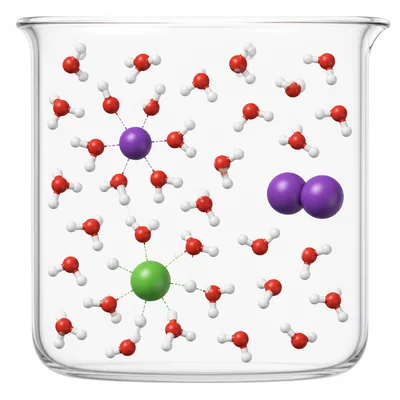
Predict which substance is more soluble in water: sodium chloride, NaCl, or iodine, I2. Explain your answer using particle attractions.
- 10
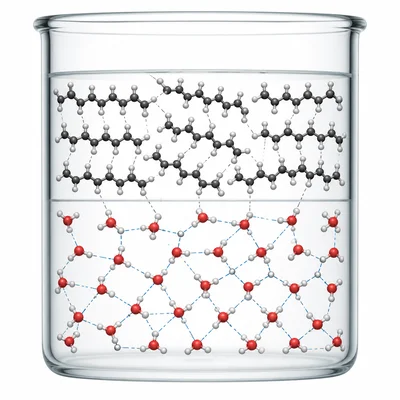
Oil and water separate into two layers when mixed. Explain this observation using polarity and intermolecular forces.
- 11
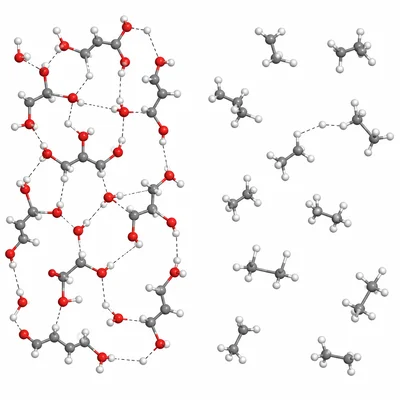
Which liquid would you expect to be more viscous at room temperature: glycerol, C3H8O3, or propane, C3H8? Explain your reasoning.
- 12
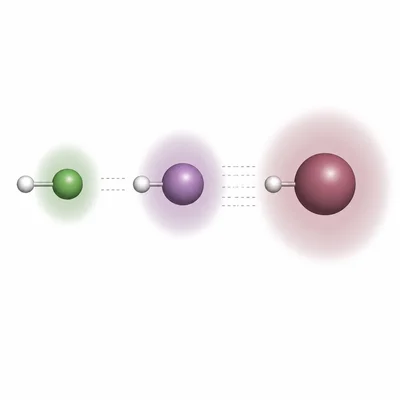
Rank HCl, HBr, and HI from lowest boiling point to highest boiling point. All three are polar molecules, but hydrogen bonding is not present. Explain your ranking.
- 13
A mystery liquid has a boiling point of 80 degrees C, while another similar-sized liquid has a boiling point of 10 degrees C. Which liquid likely has stronger intermolecular forces? Explain.
- 14
Explain why ammonia, NH3, has a higher boiling point than phosphine, PH3, even though PH3 has a greater molar mass.
- 15
A student is comparing two molecules: molecule A is nonpolar with a molar mass of 58 g/mol, and molecule B is polar with a molar mass of 30 g/mol. The student claims molecule B must always have the higher boiling point because it is polar. Evaluate the claim.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems