Gas Laws Cheat Sheet
A printable reference covering Boyle's law, Charles's law, Gay-Lussac's law, the ideal gas law, and Dalton's law for grades 10-11.
Related Tools
Related Worksheets
Related Infographics
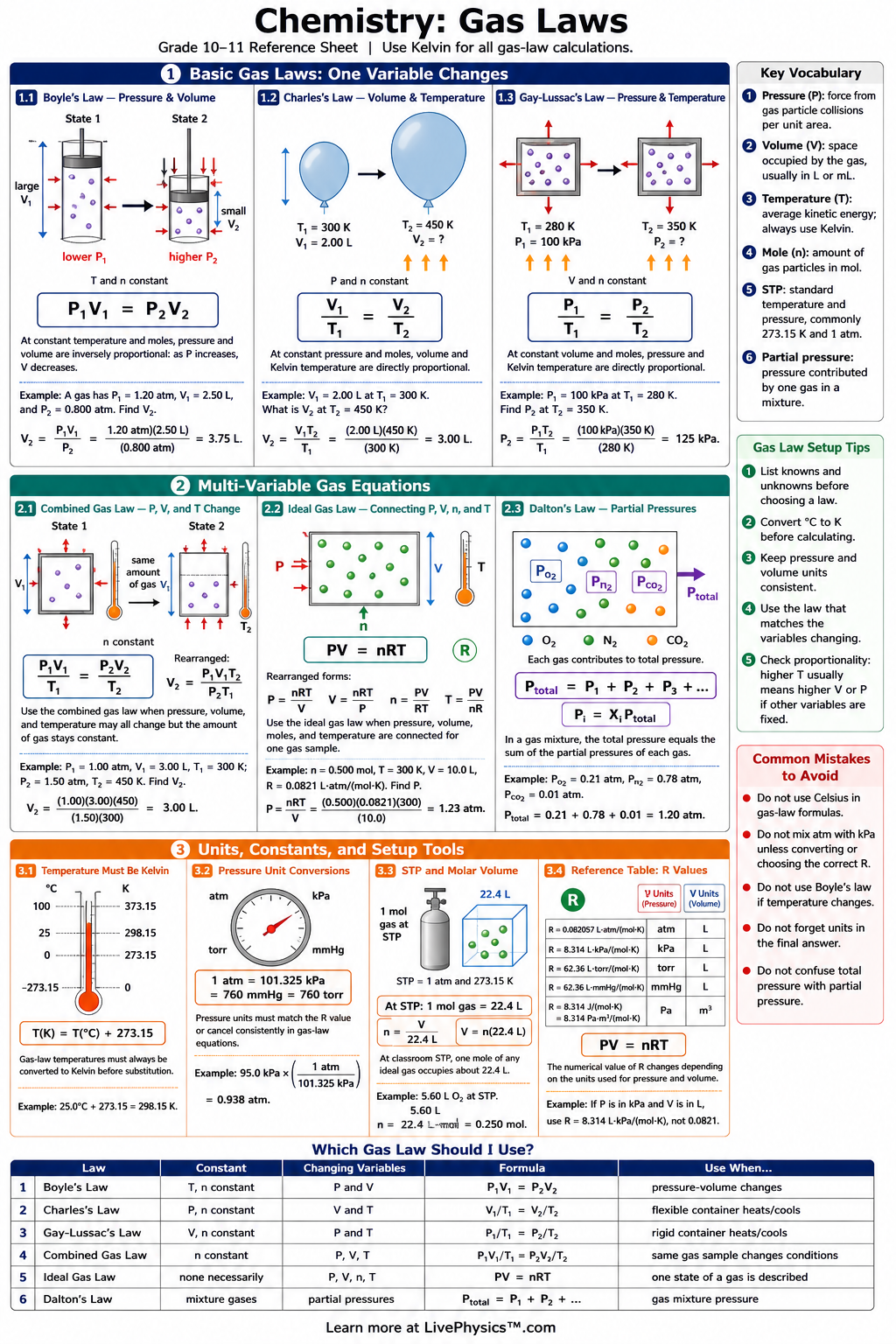
Gas laws describe how pressure, volume, temperature, and amount of gas are related. This cheat sheet helps students choose the correct equation, identify units, and solve common gas law problems. It is useful for chemistry labs, homework, and test review because gas variables often change together. The most important idea is that gases are modeled using proportional relationships. Boyle's law connects pressure and volume, Charles's law connects volume and temperature, and Gay-Lussac's law connects pressure and temperature. The combined gas law and ideal gas law bring several variables together, while Dalton's law explains mixtures of gases.
Key Facts
- Boyle's law states that pressure and volume are inversely proportional when temperature and moles stay constant: .
- Charles's law states that volume and temperature are directly proportional when pressure and moles stay constant: .
- Gay-Lussac's law states that pressure and temperature are directly proportional when volume and moles stay constant: .
- The combined gas law relates pressure, volume, and temperature for a fixed amount of gas: .
- The ideal gas law relates pressure, volume, moles, and temperature: .
- Temperature in gas law calculations must be measured in kelvins, using .
- Dalton's law of partial pressures states that the total pressure of a gas mixture is the sum of each gas pressure: .
- At standard temperature and pressure, mole of an ideal gas has a volume of about at and .
Vocabulary
- Pressure
- Pressure is the force of gas particle collisions per unit area, often measured in , , or .
- Volume
- Volume is the amount of space a gas occupies, usually measured in or .
- Kelvin
- Kelvin is the absolute temperature scale used in gas laws, where represents absolute zero.
- Mole
- A mole is an amount of substance equal to particles.
- Ideal Gas
- An ideal gas is a model gas whose particles have no volume and no intermolecular attractions.
- Partial Pressure
- Partial pressure is the pressure one gas in a mixture would exert if it occupied the container alone.
Common Mistakes to Avoid
- Using Celsius in gas law equations is wrong because proportional gas laws require absolute temperature. Always convert with before substituting.
- Mixing pressure units is wrong because equations compare or combine pressures directly. Convert all pressures to the same unit, such as or , before solving.
- Choosing Boyle's law for a temperature change is wrong because Boyle's law assumes temperature is constant. If pressure, volume, and temperature all change, use .
- Forgetting that inverse relationships move in opposite directions is wrong for Boyle's law. If volume decreases at constant temperature, pressure must increase.
- Using the wrong gas constant is wrong because must match the pressure and volume units. For example, use when pressure is in and volume is in .
Practice Questions
- 1 A gas has a volume of at . If the pressure increases to at constant temperature, what is the new volume?
- 2 A balloon contains of gas at . What volume will it have at if pressure stays constant?
- 3 How many moles of gas are in a container at and ? Use .
- 4 Explain why a sealed aerosol can may become dangerous when heated, using particle motion, pressure, and temperature.