Science: The Mole and Avogadros Number
Using the mole to count particles in chemistry
Using the mole to count particles in chemistry
Chemistry - Grade 9-12
- 1
Define a mole in chemistry and state Avogadros number.
- 2
How many atoms are in 2.0 moles of helium?
- 3
How many moles are represented by 3.011 x 10^23 molecules of oxygen gas, O2?
- 4
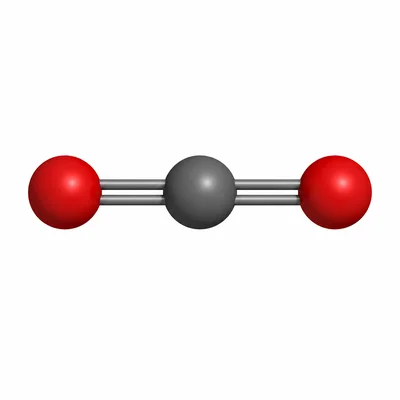
What is the molar mass of carbon dioxide, CO2?
- 5
How many grams are in 0.75 moles of sodium chloride, NaCl? Use a molar mass of 58.44 g/mol.
- 6
How many moles are in 36.0 grams of water, H2O? Use a molar mass of 18.02 g/mol.
- 7
A sample contains 1.50 moles of magnesium atoms. How many atoms are present?
- 8
A sample contains 4.50 x 10^24 atoms of aluminum. How many moles of aluminum atoms is this?
- 9
How many molecules are in 0.250 moles of methane, CH4?
- 10
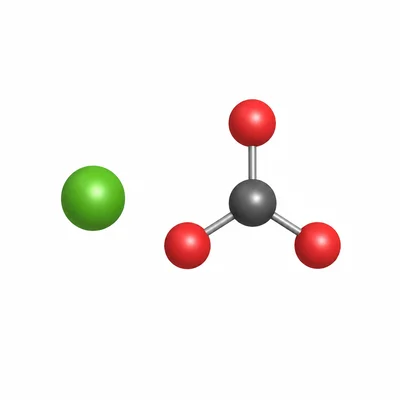
What is the molar mass of calcium carbonate, CaCO3?
- 11
How many formula units are in 3.25 moles of calcium chloride, CaCl2?
- 12
Explain why chemists use the mole instead of counting atoms or molecules one by one.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems