Enzymes and Biochemistry
Enzymes and Biochemistry
Related Tools
Related Worksheets
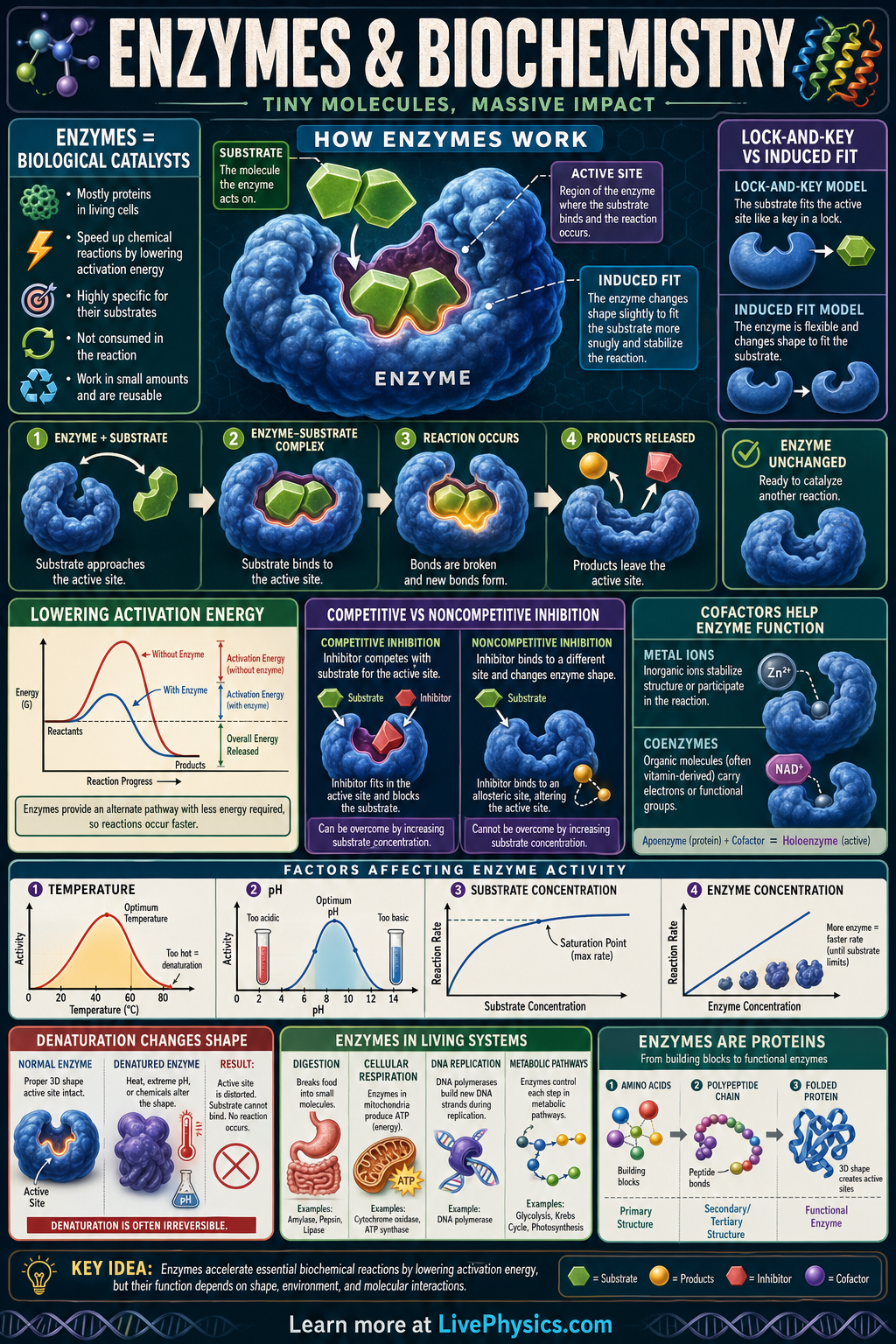
Enzymes are biological catalysts that speed up chemical reactions in living systems without being used up. They are essential for digestion, energy release, DNA replication, and many other processes that keep cells alive. By lowering the activation energy of a reaction, enzymes make important reactions happen fast enough at normal body temperatures. Understanding enzymes helps explain how metabolism works and why temperature, pH, and concentration affect life.
Each enzyme has an active site whose shape and chemical properties allow it to bind specific substrates. When the substrate binds, the enzyme stabilizes the transition state and helps break or form chemical bonds more easily. Enzyme activity depends on factors such as temperature, pH, substrate concentration, and the presence of inhibitors or activators. In biochemistry, enzymes are also studied using reaction rates, models like lock and key or induced fit, and equations such as Michaelis-Menten kinetics.
Key Facts
- Enzymes lower activation energy, Ea, which increases reaction rate without changing the overall delta G of the reaction.
- The basic enzyme reaction can be written as E + S <-> ES -> E + P.
- At low substrate concentration, reaction rate increases as substrate concentration increases, but it approaches a maximum rate, Vmax, at saturation.
- Michaelis-Menten equation: v = (Vmax[S])/(Km + [S]).
- A lower Km usually means the enzyme reaches half of Vmax at a lower substrate concentration, indicating higher apparent substrate affinity.
- Temperature and pH each have an optimum range; outside that range, enzyme structure and activity can decrease sharply due to denaturation or altered active site chemistry.
Vocabulary
- Active site
- The active site is the specific region of an enzyme where the substrate binds and the reaction is catalyzed.
- Substrate
- A substrate is the reactant molecule that an enzyme acts on during a biochemical reaction.
- Activation energy
- Activation energy is the minimum energy needed for reactants to reach the transition state and begin a reaction.
- Denaturation
- Denaturation is the loss of an enzyme's normal shape, which reduces or destroys its ability to function.
- Competitive inhibitor
- A competitive inhibitor is a molecule that reduces enzyme activity by competing with the substrate for the active site.
Common Mistakes to Avoid
- Saying enzymes are used up in reactions, which is wrong because enzymes are regenerated after catalysis and can be reused many times.
- Assuming enzymes change the overall energy released by a reaction, which is wrong because they lower activation energy but do not change delta G or reaction equilibrium.
- Thinking higher temperature always makes enzymes work better, which is wrong because activity rises only up to an optimum and then often falls as the enzyme denatures.
- Confusing substrate specificity with absolute rigidity, which is wrong because many enzymes follow induced fit and change shape slightly when the substrate binds.
Practice Questions
- 1 An enzyme-catalyzed reaction has Vmax = 120 micromoles per minute and Km = 3.0 mM. Calculate the reaction rate v when the substrate concentration [S] is 3.0 mM using v = (Vmax[S])/(Km + [S]).
- 2 A reaction proceeds at 18 units per minute without an inhibitor and 10 units per minute with an inhibitor. What is the percent decrease in reaction rate caused by the inhibitor?
- 3 A student measures enzyme activity at pH 2, pH 7, and pH 11 and finds the highest rate at pH 7. Explain what this suggests about the enzyme's active site and why activity drops at the other pH values.