Types of Chemical Reactions
Types of Chemical Reactions
Related Tools
Related Worksheets
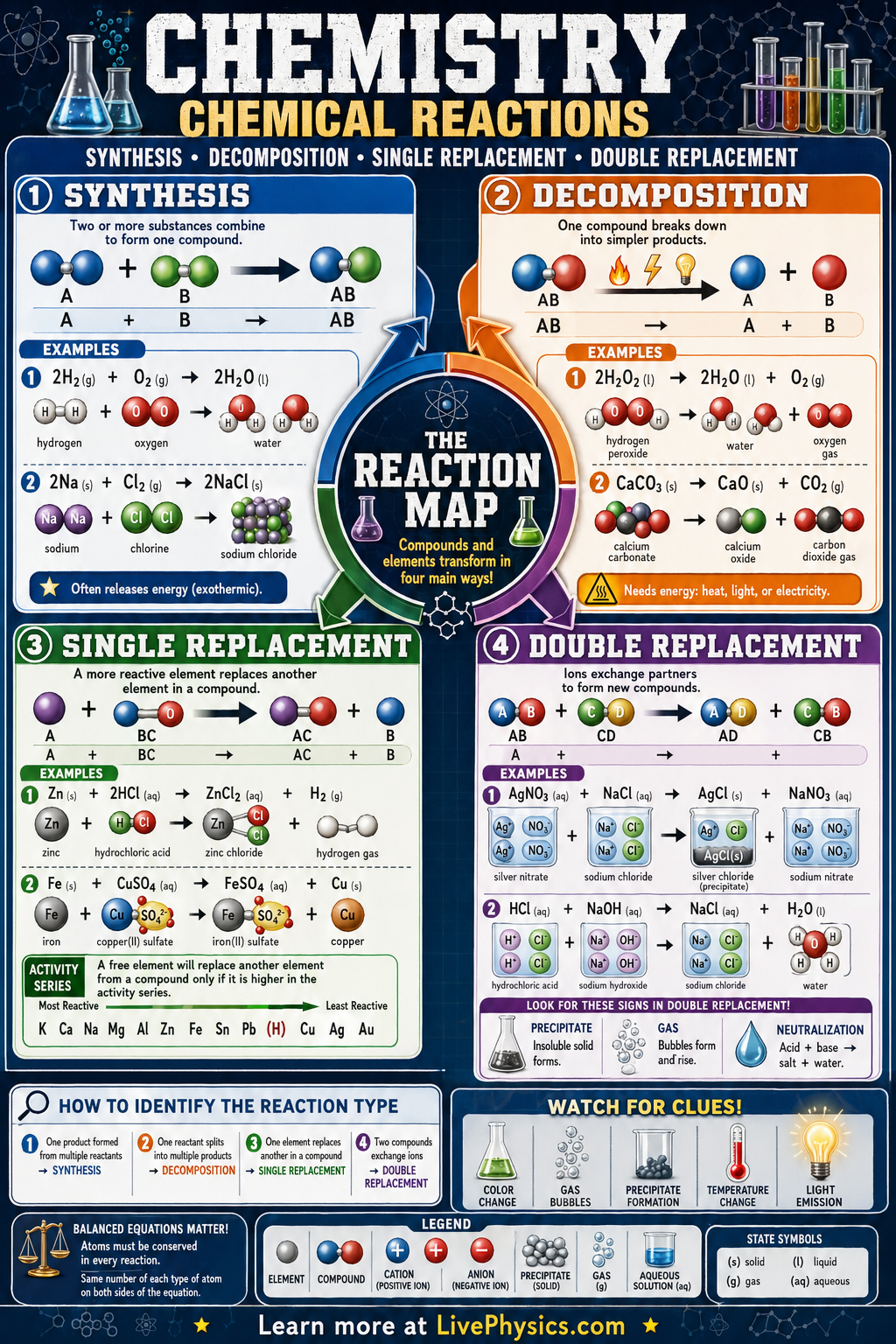
Chemical reactions rearrange atoms to form new substances, and chemists classify many of them into common patterns. Four important types are synthesis, decomposition, single replacement, and double replacement. Learning these patterns helps students predict products, balance equations, and connect symbolic equations to real laboratory changes. These reaction types also appear in industry, biology, and everyday materials.
Each reaction type follows a recognizable structure based on how atoms or ions are reorganized. In synthesis, simpler substances combine to make a more complex product, while decomposition breaks one compound into simpler parts. In single replacement, one element displaces another in a compound, and in double replacement, ions swap partners between two compounds. Recognizing these patterns makes it easier to analyze reactivity, identify precipitates or gases, and understand why some reactions occur while others do not.
Key Facts
- Synthesis reaction pattern:
- Decomposition reaction pattern:
- Single replacement reaction pattern:
- Double replacement reaction pattern:
- Chemical equations must obey conservation of mass, so the number of each type of atom is the same on both sides.
- Single replacement occurs only if the free element is more reactive than the element it replaces, often checked with an activity series.
Vocabulary
- Synthesis reaction
- A reaction in which two or more simpler substances combine to form one compound.
- Decomposition reaction
- A reaction in which one compound breaks apart into two or more simpler substances.
- Single replacement reaction
- A reaction in which one element replaces another element in a compound.
- Double replacement reaction
- A reaction in which the positive and negative ions of two compounds exchange partners.
- Activity series
- A ranking of elements by reactivity that helps predict whether a single replacement reaction will occur.
Common Mistakes to Avoid
- Assuming every element can replace any other element in a single replacement reaction, which is wrong because the free element must be more reactive than the one in the compound.
- Forgetting to balance the equation after identifying the reaction type, which is wrong because atoms must be conserved in every chemical reaction.
- Mixing up synthesis and decomposition, which is wrong because synthesis combines substances into one product while decomposition starts with one reactant and splits it apart.
- Swapping subscripts when writing double replacement products, which is wrong because subscripts belong to the ions and should not be changed unless balancing requires coefficients.
Practice Questions
- 1 Classify the reaction and balance it: .
- 2 Classify the reaction and complete the products, then balance: ?
- 3 A student mixes two ionic compounds in water and sees a solid form after the ions exchange partners. Explain why this is classified as a double replacement reaction rather than a single replacement reaction.