Chemical Reactions: Signs and Types
Identifying evidence of reactions and common reaction types
Identifying evidence of reactions and common reaction types
Chemistry - Grade 6-8
- 1

A student mixes vinegar and baking soda in a cup. The mixture bubbles and foams. What sign of a chemical reaction is shown, and what does it tell you?
- 2

When a piece of iron is left outside for many weeks, reddish-brown rust forms on its surface. Is this a physical change or a chemical reaction? Explain your answer.
- 3
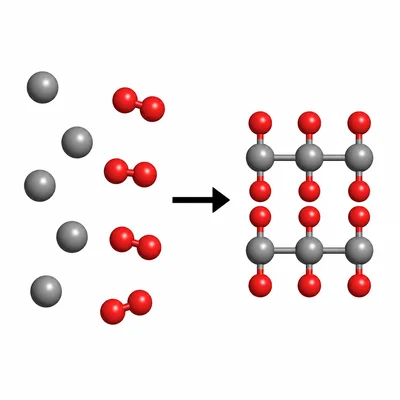
Classify this reaction as synthesis, decomposition, single replacement, double replacement, or combustion: magnesium plus oxygen forms magnesium oxide.
- 4
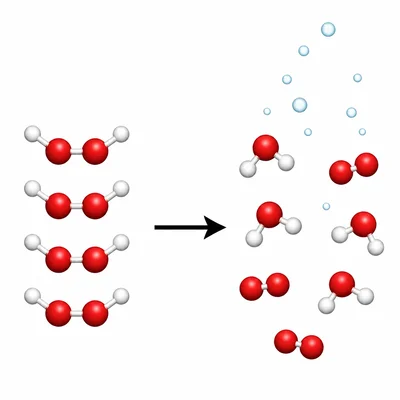
Classify this reaction as synthesis, decomposition, single replacement, double replacement, or combustion: hydrogen peroxide breaks down into water and oxygen gas.
- 5

A candle burns in oxygen and produces carbon dioxide, water vapor, heat, and light. What type of reaction is this? Give one piece of evidence that a chemical reaction occurred.
- 6

Two clear liquids are mixed in a test tube. A cloudy white solid appears and settles at the bottom. What sign of a chemical reaction is shown?
- 7

Classify this reaction as synthesis, decomposition, single replacement, double replacement, or combustion: zinc metal reacts with hydrochloric acid to form zinc chloride and hydrogen gas.
- 8
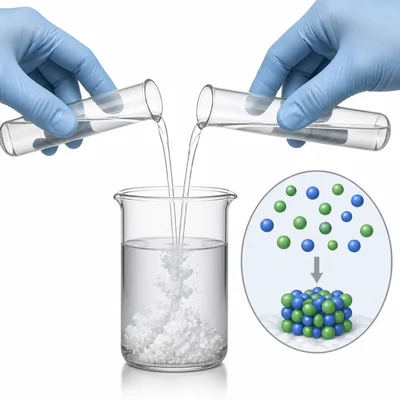
Classify this reaction as synthesis, decomposition, single replacement, double replacement, or combustion: silver nitrate reacts with sodium chloride to form silver chloride and sodium nitrate.
- 9

A student mixes two room-temperature solutions. The cup becomes noticeably warmer, but no flame appears. What sign of a chemical reaction is shown?
- 10

A glow stick is bent and shaken. It begins to glow without being plugged in or heated. What sign of a chemical reaction is shown?
- 11

Decide whether this change is most likely physical or chemical: An ice cube melts into liquid water. Explain your answer.
- 12

Decide whether this change is most likely physical or chemical: A piece of paper turns black and gives off smoke when it burns. Explain your answer.
- 13

Look at this word equation: calcium carbonate produces calcium oxide plus carbon dioxide. What type of reaction is it, and what sign might you observe?
- 14

A blue copper chloride solution is added to a piece of aluminum foil. After a few minutes, the foil looks darker and a reddish solid appears. What evidence suggests a chemical reaction occurred?
- 15
Write one original example of a chemical reaction and identify at least two signs that would show the reaction happened.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems