Chemistry: Conservation of Mass in Reactions
Tracking atoms and mass before and after chemical reactions
Tracking atoms and mass before and after chemical reactions
Chemistry - Grade 6-8
- 1
State the law of conservation of mass in your own words.
- 2
In a closed container, 12 grams of hydrogen reacts with 96 grams of oxygen to form water. What is the total mass of water produced?
- 3

A student mixes two clear liquids in a sealed bag. The mass before mixing is 145 grams. After a reaction occurs, the mass is still 145 grams. Explain why the mass did not change.
- 4

A reaction in an open beaker starts with 50 grams of vinegar and 5 grams of baking soda. After the reaction, the beaker and contents have a mass of 52 grams. Why is the measured mass less than 55 grams?
- 5
A closed container holds 30 grams of reactant A and 18 grams of reactant B. They react completely to form one product. What is the mass of the product?
- 6
Magnesium burns in oxygen to form magnesium oxide. If 24 grams of magnesium combines with 16 grams of oxygen, what mass of magnesium oxide forms?
- 7
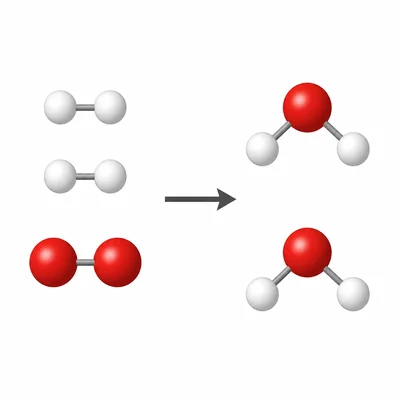
Count the atoms in this reaction: 2H2 + O2 -> 2H2O. How many hydrogen atoms and oxygen atoms are on each side?
- 8
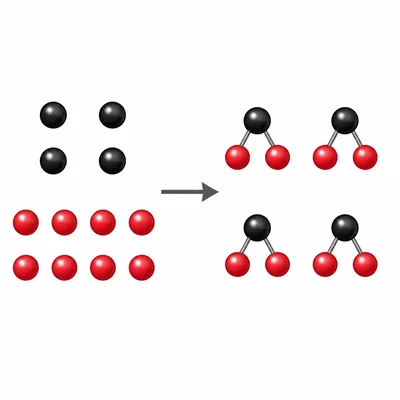
A diagram shows 4 carbon atoms and 8 oxygen atoms before a reaction. After the reaction, the products must contain how many carbon atoms and oxygen atoms in total?
- 9

A sealed jar contains iron and oxygen. The total mass before rust forms is 200 grams. What should the total mass be after rust forms inside the sealed jar?
- 10
A science class records the mass of a closed reaction system as 86.4 grams before a reaction. After the reaction, the mass is 86.1 grams. Give one possible reason for the small difference.
- 11

A chemical reaction has 75 grams of reactants. The products are 30 grams of solid and an unknown mass of gas. In a closed system, what is the mass of the gas?
- 12
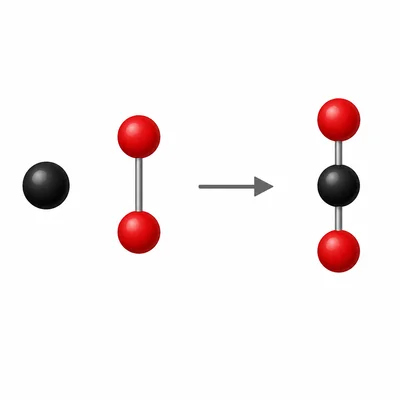
The reaction C + O2 -> CO2 represents carbon reacting with oxygen to form carbon dioxide. Is the number of atoms conserved in this equation? Explain.
- 13

A student says, 'The candle wax disappeared when the candle burned, so mass was destroyed.' Explain why this statement is not correct.
- 14

A tablet is dropped into water in a sealed bottle. The bottle expands slightly as bubbles form, but the cap stays on. The mass before the reaction is 132 grams. What is the mass after the reaction, and why?
- 15
A reaction uses 10 grams of substance X and 25 grams of substance Y. It produces 20 grams of substance Z and 15 grams of substance W. Does this data support the law of conservation of mass? Explain.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems