Science: Types of Chemical Reactions
Identifying synthesis, decomposition, single replacement, double replacement, and combustion
Identifying synthesis, decomposition, single replacement, double replacement, and combustion
Chemistry - Grade 6-8
- 1

A reaction combines iron and sulfur to make iron sulfide: Fe + S -> FeS. What type of chemical reaction is this?
- 2
Calcium carbonate breaks down when heated: CaCO3 -> CaO + CO2. What type of chemical reaction is this?
- 3
Zinc reacts with hydrochloric acid: Zn + 2HCl -> ZnCl2 + H2. What type of chemical reaction is this?
- 4
Silver nitrate reacts with sodium chloride: AgNO3 + NaCl -> AgCl + NaNO3. What type of chemical reaction is this?
- 5
Methane burns in oxygen: CH4 + 2O2 -> CO2 + 2H2O. What type of chemical reaction is this?
- 6
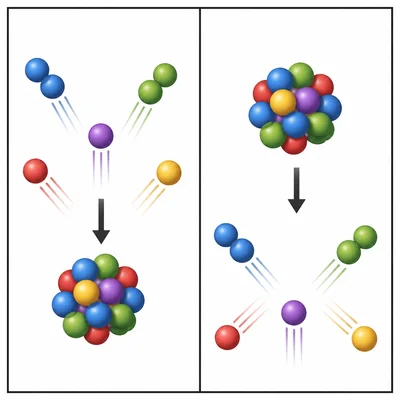
Describe what makes a synthesis reaction different from a decomposition reaction.
- 7
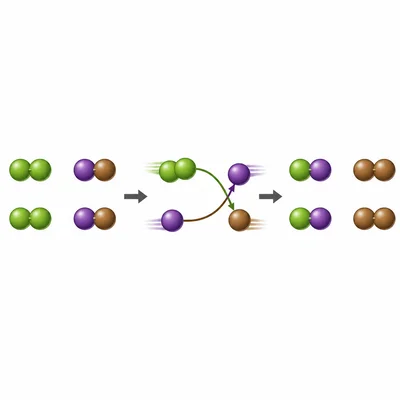
In the reaction Cl2 + 2KBr -> 2KCl + Br2, what type of reaction occurs, and which element is replaced?
- 8
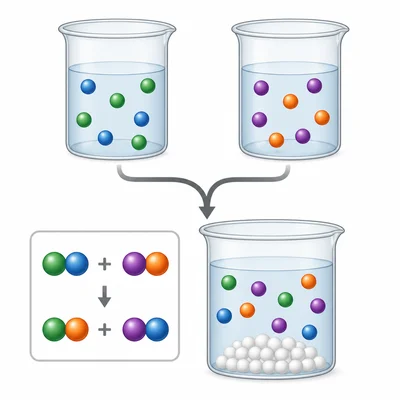
In the reaction BaCl2 + Na2SO4 -> BaSO4 + 2NaCl, what type of reaction occurs?
- 9
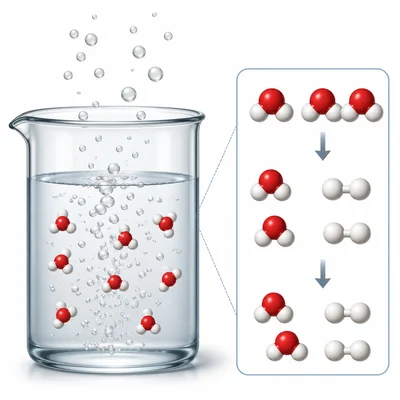
Hydrogen peroxide breaks down into water and oxygen: 2H2O2 -> 2H2O + O2. What type of reaction is this?
- 10
Magnesium burns in oxygen to form magnesium oxide: 2Mg + O2 -> 2MgO. Name the reaction type and explain your choice.
- 11

A student says that every reaction with oxygen is combustion. Is that statement always correct? Explain.
- 12

Classify this reaction: 2KClO3 -> 2KCl + 3O2. Then explain what happens to the reactant.
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems