Chemistry
Colligative Properties
Colligative Properties
Related Tools
Related Labs
Related Worksheets
Related Cheat Sheets
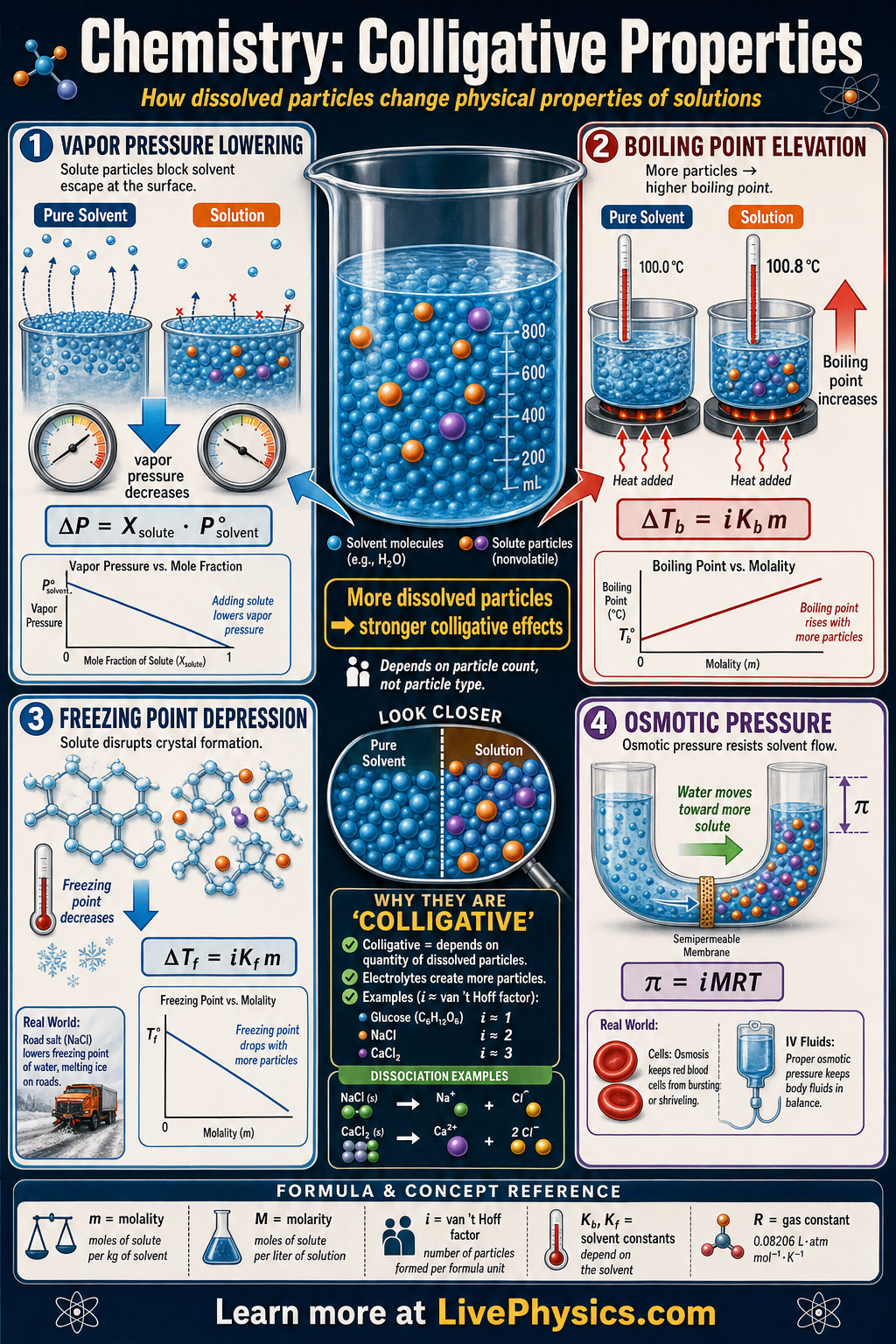
Colligative properties are changes in a solvent that depend on how many solute particles are dissolved, not on the chemical identity of those particles. They explain why salt melts ice, why antifreeze protects engines, and why concentrated solutions boil and freeze at different temperatures than pure liquids. These effects are especially important in chemistry, biology, environmental science, and engineering because real solutions rarely behave exactly like pure substances.
Key Facts
- Colligative properties depend on the number of dissolved particles, not their type.
- Boiling point elevation: ΔTb = iKb m
- Freezing point depression: ΔTf = iKf m
- Osmotic pressure: π = iMRT
- Vapor pressure lowering follows Raoult's law for ideal solutions: Psolution = Xsolvent P°solvent
- Molality is m = moles of solute per kilogram of solvent and is used because it does not change with temperature.
Vocabulary
- Colligative property
- A solution property that depends on the concentration of dissolved particles rather than their chemical identity.
- Molality
- A concentration unit equal to moles of solute divided by kilograms of solvent.
- Van't Hoff factor
- The number i that represents how many particles one formula unit of solute produces in solution.
- Osmosis
- The movement of solvent through a semipermeable membrane from a lower solute concentration to a higher solute concentration.
- Vapor pressure
- The pressure exerted by vapor above a liquid when evaporation and condensation are balanced.
Common Mistakes to Avoid
- Using molarity instead of molality for boiling and freezing point calculations is wrong because ΔTb and ΔTf formulas require moles of solute per kilogram of solvent.
- Forgetting the Van't Hoff factor gives answers that are too small for ionic solutes because compounds like NaCl or CaCl2 separate into multiple particles in water.
- Thinking solute identity never matters in any way is wrong because colligative formulas depend mainly on particle number, but real solutions can deviate when particles attract, pair up, or react.
- Adding the freezing point change instead of subtracting it is wrong because solutes lower the freezing point, so the new freezing point is Tpure - ΔTf.
Practice Questions
- 1 A solution is made by dissolving 0.50 mol of glucose in 2.00 kg of water. If Kf for water is 1.86 °C kg/mol and i = 1, what is the freezing point of the solution?
- 2 What is the boiling point of a 0.75 m NaCl solution in water if Kb = 0.512 °C kg/mol and the ideal Van't Hoff factor is i = 2?
- 3 Two solutions have the same molality: glucose in water and calcium chloride in water. Which solution should have the greater freezing point depression, and why?