States of Matter
Solid, Liquid, and Gas
Related Tools
Related Labs
Related Worksheets
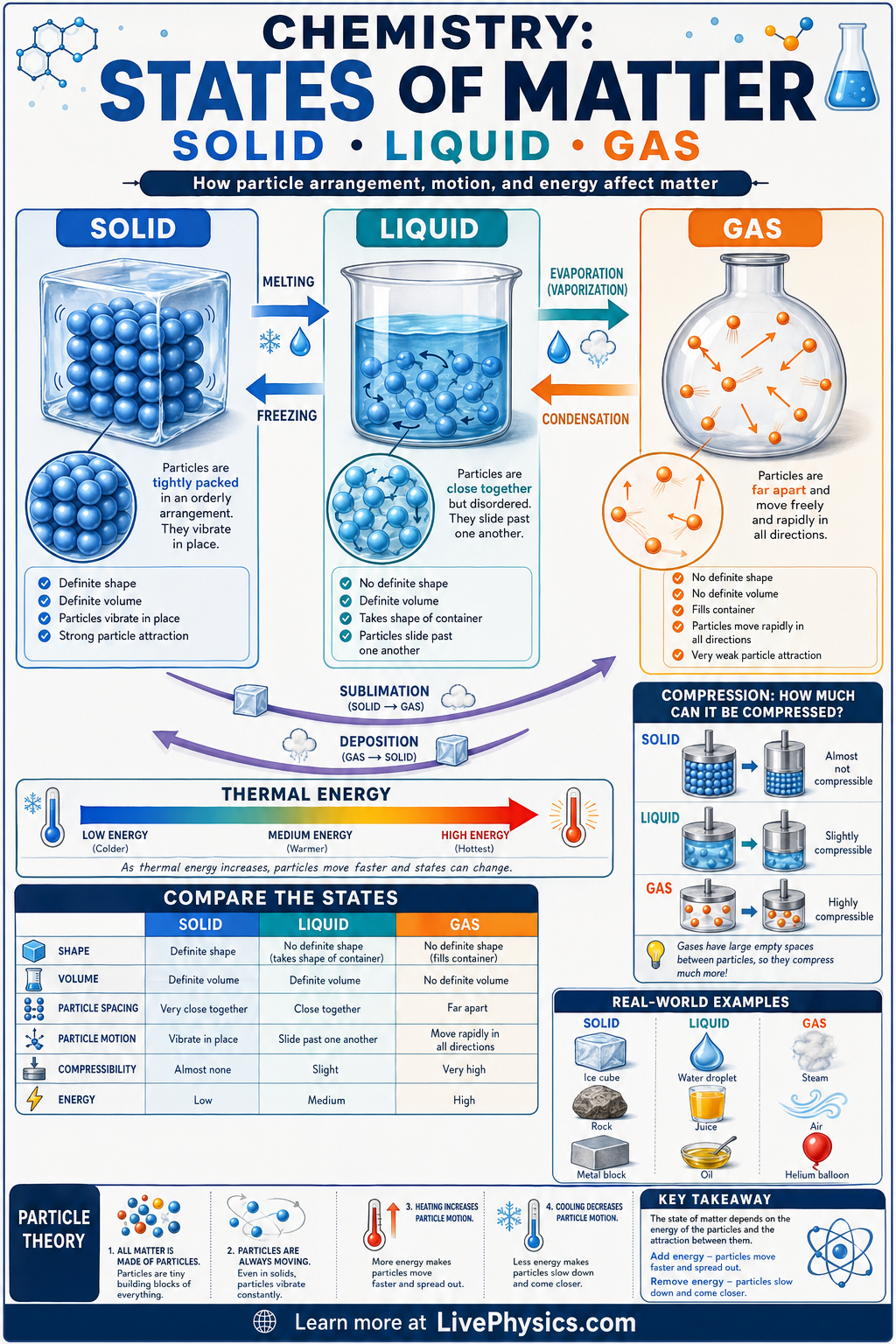
Matter is commonly grouped into three familiar states: solid, liquid, and gas. These states help explain why ice keeps its shape, water flows, and steam spreads to fill a room. The differences come from how particles are arranged and how strongly they attract each other. Understanding states of matter is a foundation for chemistry, physics, and everyday phenomena like melting, boiling, and condensation.
In a solid, particles are packed closely and mainly vibrate in place, so the material has a fixed shape and volume. In a liquid, particles remain close together but can slide past one another, giving a fixed volume but no fixed shape. In a gas, particles are far apart and move freely, so gases have neither fixed shape nor fixed volume. Changes between states happen when energy is added or removed, changing particle motion and intermolecular attraction.
Key Facts
- Solid: fixed shape and fixed volume.
- Liquid: no fixed shape, but fixed volume.
- Gas: no fixed shape and no fixed volume.
- Heating generally increases particle kinetic energy.
- Cooling generally decreases particle kinetic energy.
Vocabulary
- Solid
- A state of matter with particles packed closely together that keeps a fixed shape and a fixed volume.
- Liquid
- A state of matter with particles close together that flows and takes the shape of its container while keeping a fixed volume.
- Gas
- A state of matter with particles far apart that moves freely and expands to fill its container.
- Intermolecular forces
- These are attractive forces between particles that help determine whether a substance is solid, liquid, or gas.
- Phase change
- A phase change is the transition of a substance from one state of matter to another, such as melting or boiling.
Common Mistakes to Avoid
- Thinking gases have no mass, which is wrong because gas particles are still matter and contribute to mass and density.
- Assuming liquids are highly compressible, which is wrong because their particles are already close together and cannot be squeezed much closer.
- Believing particles in a solid do not move at all, which is wrong because they vibrate around fixed positions.
- Confusing evaporation with boiling, which is wrong because evaporation happens at the surface at many temperatures while boiling occurs throughout the liquid at its boiling point.
Practice Questions
- 1 A sample of water has a mass of 200 g and a volume of 250 cm^3. Calculate its density using .
- 2 A gas in a syringe occupies 40 mL. It is compressed to 25 mL while the amount of gas stays the same. Does the gas have a fixed volume, and what property of gases does this demonstrate?
- 3 Why does a solid usually keep its own shape while a gas spreads out to fill the entire container? Explain using particle arrangement and motion.