Chemistry: Ionic Bonding and Lattice Structures
Electron transfer, crystal lattices, and properties of ionic compounds
Electron transfer, crystal lattices, and properties of ionic compounds
Chemistry - Grade 9-12
- 1
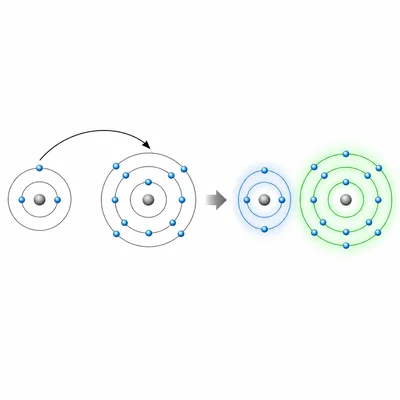
Explain how an ionic bond forms between sodium and chlorine atoms. Include what happens to the electrons and name the ions that form.
- 2
Write the correct chemical formula for the ionic compound formed from magnesium ions, Mg2+, and oxide ions, O2-. Explain why this formula is neutral.
- 3
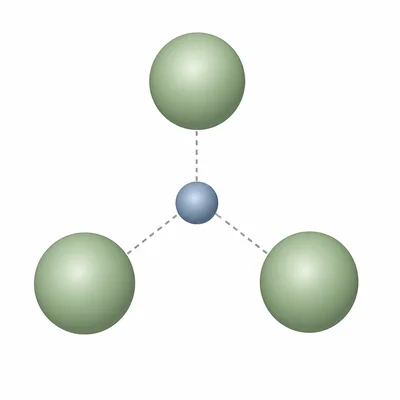
Write the correct chemical formula for the ionic compound formed from aluminum ions, Al3+, and chloride ions, Cl-. Explain how you balanced the charges.
- 4
A compound has the formula CaF2. Identify the ions present and state the charge on each ion.
- 5
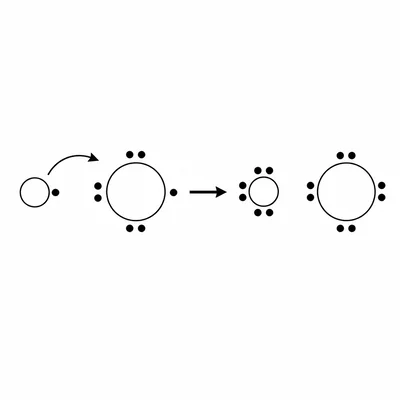
Draw or describe the Lewis dot process for forming lithium fluoride, LiF. State which atom loses an electron and which atom gains an electron.
- 6
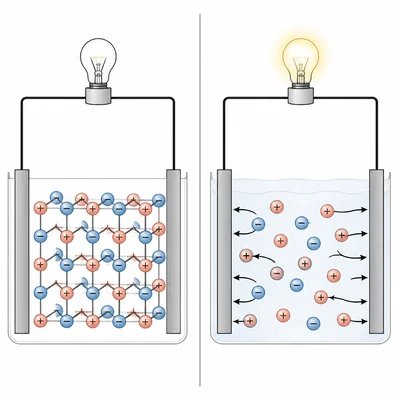
Explain why solid sodium chloride does not conduct electricity, but molten sodium chloride does conduct electricity.
- 7
Compare the lattice energy of NaCl and MgO. Which compound would you expect to have the larger lattice energy, and why?
- 8
Rank these ionic compounds from lowest to highest expected melting point: NaF, MgO, KBr. Explain your ranking using ion charge and ion size.
- 9
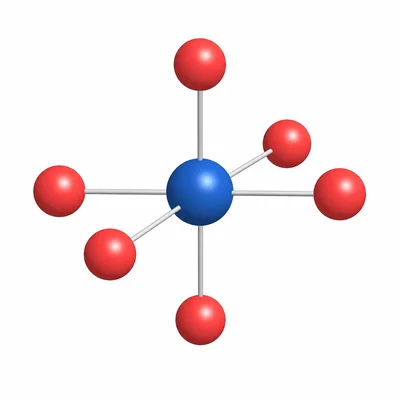
In a sodium chloride crystal lattice, each Na+ ion is surrounded by 6 Cl- ions, and each Cl- ion is surrounded by 6 Na+ ions. What is the coordination number for each ion?
- 10
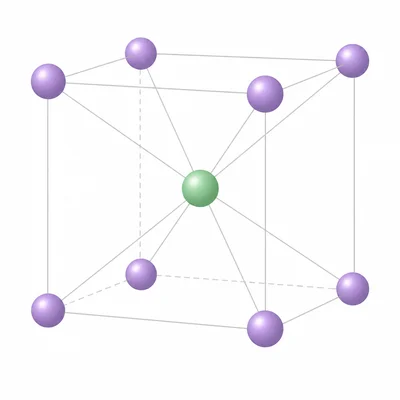
Cesium chloride has a crystal structure in which each Cs+ ion is surrounded by 8 Cl- ions. How does this coordination number compare with sodium chloride, and what does it tell you about the lattice arrangement?
- 11
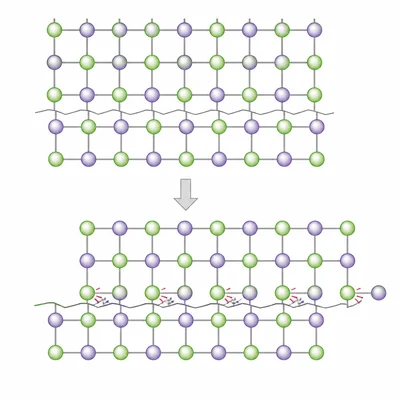
Explain why ionic compounds are usually brittle rather than flexible.
- 12
A student says, "Ionic compounds are made of molecules." Correct this statement using sodium chloride as an example.
- 13
Write the formula and name of the compound formed from potassium ions and sulfate ions, SO4 2-. Explain how the charges are balanced.
- 14
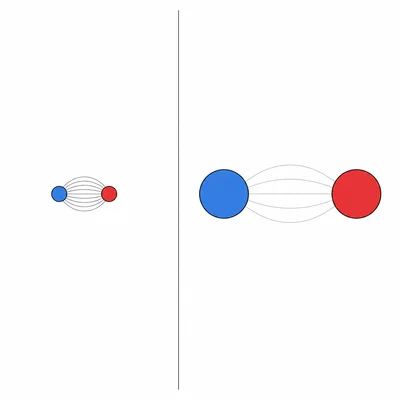
Use Coulomb's law ideas to explain why LiF generally has stronger ionic attractions than KI.
- 15
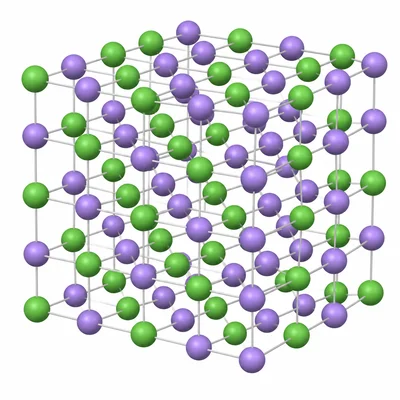
A diagram shows alternating positive and negative ions arranged in a repeating pattern. Describe two features that identify the structure as an ionic lattice.
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems