Chemistry: Kinetics: Reaction Rates and Activation Energy
Calculating reaction rates and explaining energy barriers
Calculating reaction rates and explaining energy barriers
Chemistry - Grade 9-12
- 1
A reactant has a concentration of 0.80 M at 0 seconds and 0.20 M at 30 seconds. Calculate the average rate of disappearance of the reactant.
- 2
A product concentration increases from 0.00 M to 0.45 M in 15 seconds. Calculate the average rate of formation of the product.
- 3
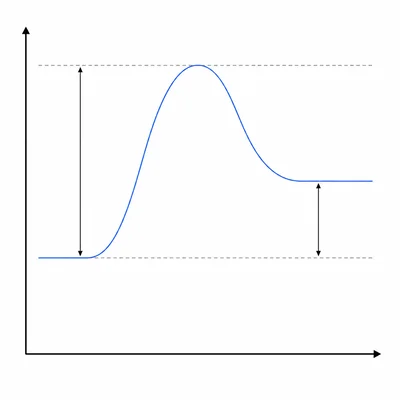
In an energy diagram, the reactants are at 40 kJ, the peak of the curve is at 125 kJ, and the products are at 70 kJ. Find the activation energy for the forward reaction and state whether the reaction is endothermic or exothermic.
- 4
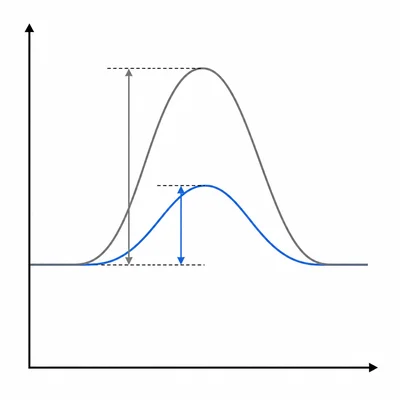
Explain how a catalyst changes the rate of a chemical reaction.
- 5
List three factors that can increase the rate of a chemical reaction and briefly explain why each one works.
- 6

A solid chunk of calcium carbonate reacts with hydrochloric acid. The same mass of powdered calcium carbonate reacts with the same acid. Which form reacts faster, and why?
- 7
The concentration of reactant A is 0.100 M at 0 seconds and 0.060 M at 20 seconds. Calculate the average rate of disappearance of A during this time interval.
- 8
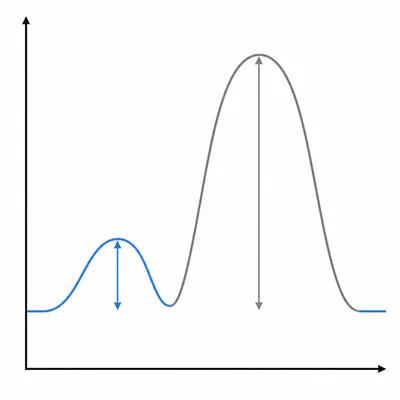
Reaction 1 has an activation energy of 35 kJ/mol. Reaction 2 has an activation energy of 85 kJ/mol. If both reactions are run at the same temperature and have similar collision frequencies, which reaction is faster? Explain your reasoning.
- 9
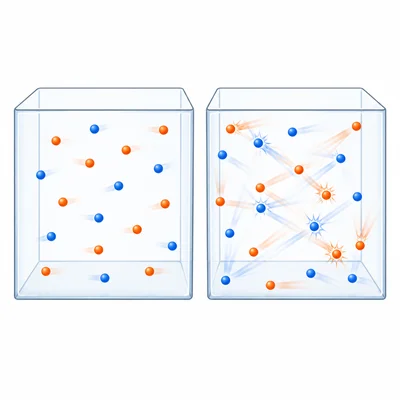
Explain why increasing temperature usually increases the rate of a reaction.
- 10
Initial rate data show that doubling the concentration of A doubles the reaction rate, while doubling the concentration of B makes the reaction rate four times larger. Write the rate law for the reaction in terms of A and B.
- 11
For a reaction with rate law rate = k[A]^2, the rate has units of M/s and concentration has units of M. Determine the units of the rate constant k.
- 12
Food spoils more slowly in a refrigerator than at room temperature. Use reaction kinetics to explain why.
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems