Science: Laws of Thermodynamics
Understanding energy, entropy, and thermal systems
Understanding energy, entropy, and thermal systems
Physics - Grade 9-12
- 1
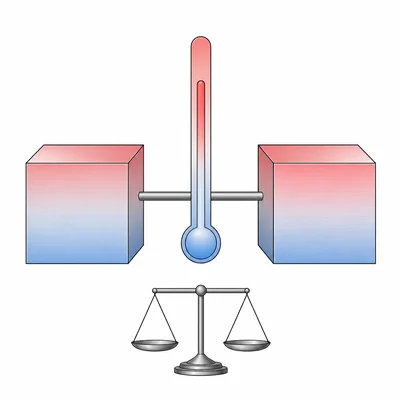
State the zeroth law of thermodynamics in your own words and explain what it means for measuring temperature.
- 2
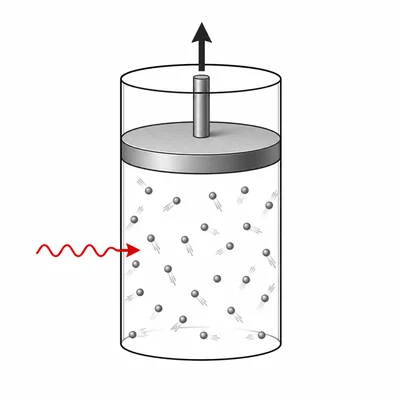
A gas in a cylinder absorbs 250 J of heat and does 90 J of work on its surroundings. Use the first law of thermodynamics to find the change in internal energy of the gas.
- 3

A metal spoon is placed in a cup of hot soup. Identify the direction of thermal energy transfer and explain which law of thermodynamics helps define when transfer stops.
- 4
Describe the first law of thermodynamics and explain how it is related to conservation of energy.
- 5
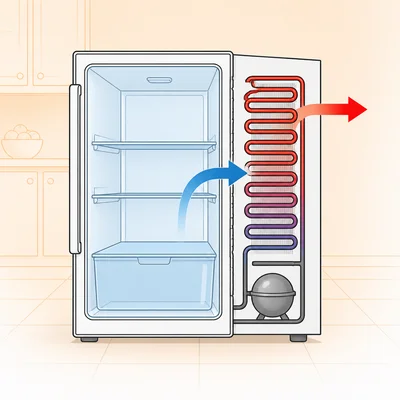
A refrigerator removes heat from its cold interior and releases it into a warmer kitchen. Explain why this process does not violate the second law of thermodynamics.
- 6
What does the second law of thermodynamics say about entropy in an isolated system?
- 7
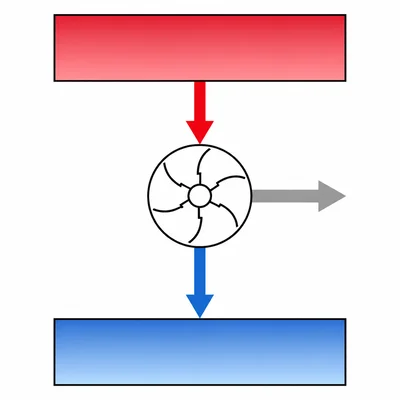
A heat engine takes in 600 J of heat from a hot reservoir and releases 420 J to a cold reservoir. How much work does the engine do?
- 8
Explain why no heat engine can be 100 percent efficient according to the second law of thermodynamics.
- 9
State the third law of thermodynamics and explain what it says about reaching absolute zero.
- 10
A student says that if a process decreases the entropy of one object, then the second law is broken. Explain why this statement is incorrect.
- 11
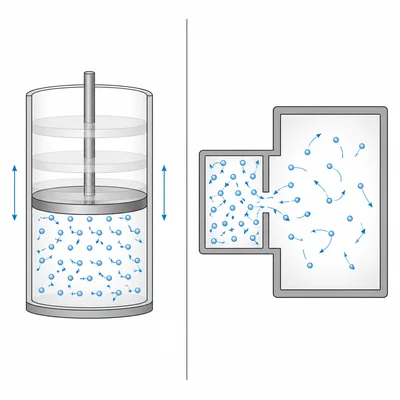
Compare a reversible process and an irreversible process in terms of entropy and energy transfer.
- 12
Give one real-world example for each of the following: the zeroth law, the first law, and the second law of thermodynamics.
Related Tools
Related Infographics
Related Cheat Sheets
More Physics Worksheets
Forces & Motion
Grade 6-8 · 8 problems
Science: Energy Thermodynamics and Heat Transfer
Grade 9-12 · 15 problems
Science: Light and Sound
Grade 4-5 · 15 problems
Science: Newtons Laws of Motion
Grade 9-12 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems