Air Is Something
Invisible but Real Matter
Related Tools
Related Labs
Related Worksheets
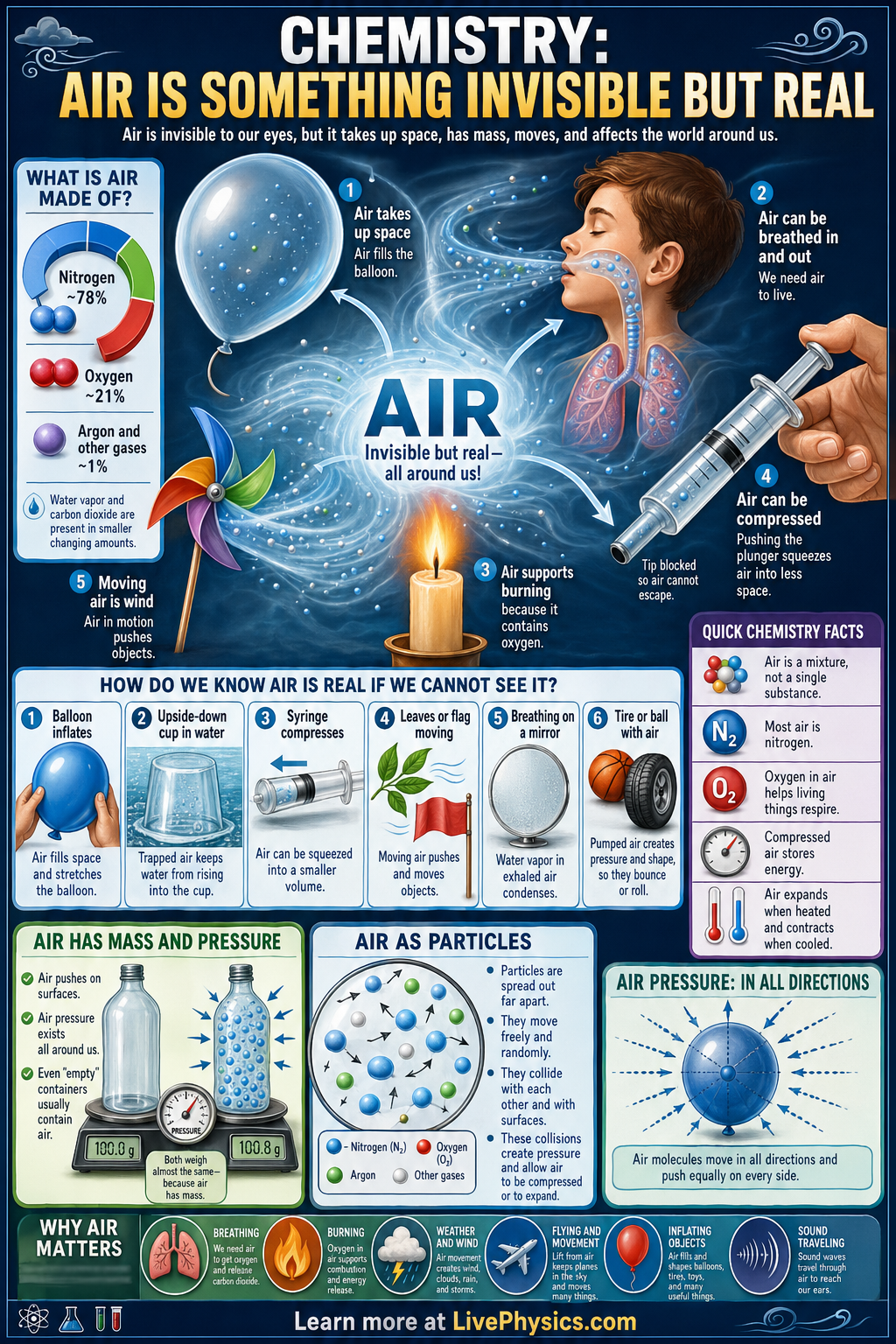
Air seems empty because we usually cannot see it, but it is a real form of matter that surrounds us and fills the space around objects. It has mass, takes up space, and pushes on surfaces as air pressure. We notice air through its effects, such as inflating a balloon, moving leaves, carrying sound, and helping living things breathe. Understanding air helps explain weather, respiration, combustion, and many everyday tools.
Air is a mixture of gases, mainly nitrogen and oxygen, with small amounts of argon, carbon dioxide, and water vapor. Its particles are always moving, so air can flow, spread out, and be compressed into a smaller volume. Heating air makes its particles move faster and usually spread farther apart, while cooling air makes them move less and often contract. Air also takes part in chemical and physical processes, including burning in a candle flame and pressure changes in pumps and syringes.
Key Facts
- Air is matter because it has mass and occupies volume.
- Earth's air is about 78% , 21% , and about 1% other gases.
- Pressure =
- For a gas at constant temperature,
- Heating a gas generally increases particle motion and can increase volume or pressure.
- Oxygen in air supports combustion, while carbon dioxide does not.
Vocabulary
- Air pressure
- Air pressure is the force per unit area caused by air particles colliding with surfaces.
- Compression
- Compression is the process of squeezing a gas into a smaller volume.
- Volume
- Volume is the amount of space that a substance or object occupies.
- Combustion
- Combustion is a chemical reaction in which a substance burns, usually using oxygen and releasing energy.
- Gas particles
- Gas particles are tiny atoms or molecules that move freely and spread out to fill their container.
Common Mistakes to Avoid
- Thinking air is nothing, which is wrong because air has mass, occupies space, and exerts pressure that can be measured.
- Assuming gases do not count as matter, which is wrong because gases are made of particles just like solids and liquids.
- Believing a syringe gets harder to push only because of the plastic, which is wrong because trapped air is being compressed and its pressure increases.
- Saying a candle burns because of the wax alone, which is wrong because combustion needs oxygen from the surrounding air to continue.
Practice Questions
- 1 A balloon contains 2.0 L of air. If the air is compressed to 0.50 L at constant temperature, and the initial pressure is 1.0 atm, what is the new pressure?
- 2 A flat basketball and pump contain 300 mL of air. After pumping, the air is compressed to 100 mL at the same temperature. If the starting pressure is 101 kPa, calculate the final pressure.
- 3 A candle is covered with a jar and goes out after a short time. Explain what this shows about air and why the flame does not keep burning.