Electrochemical Cells
Electrochemical Cells
Related Tools
Related Worksheets
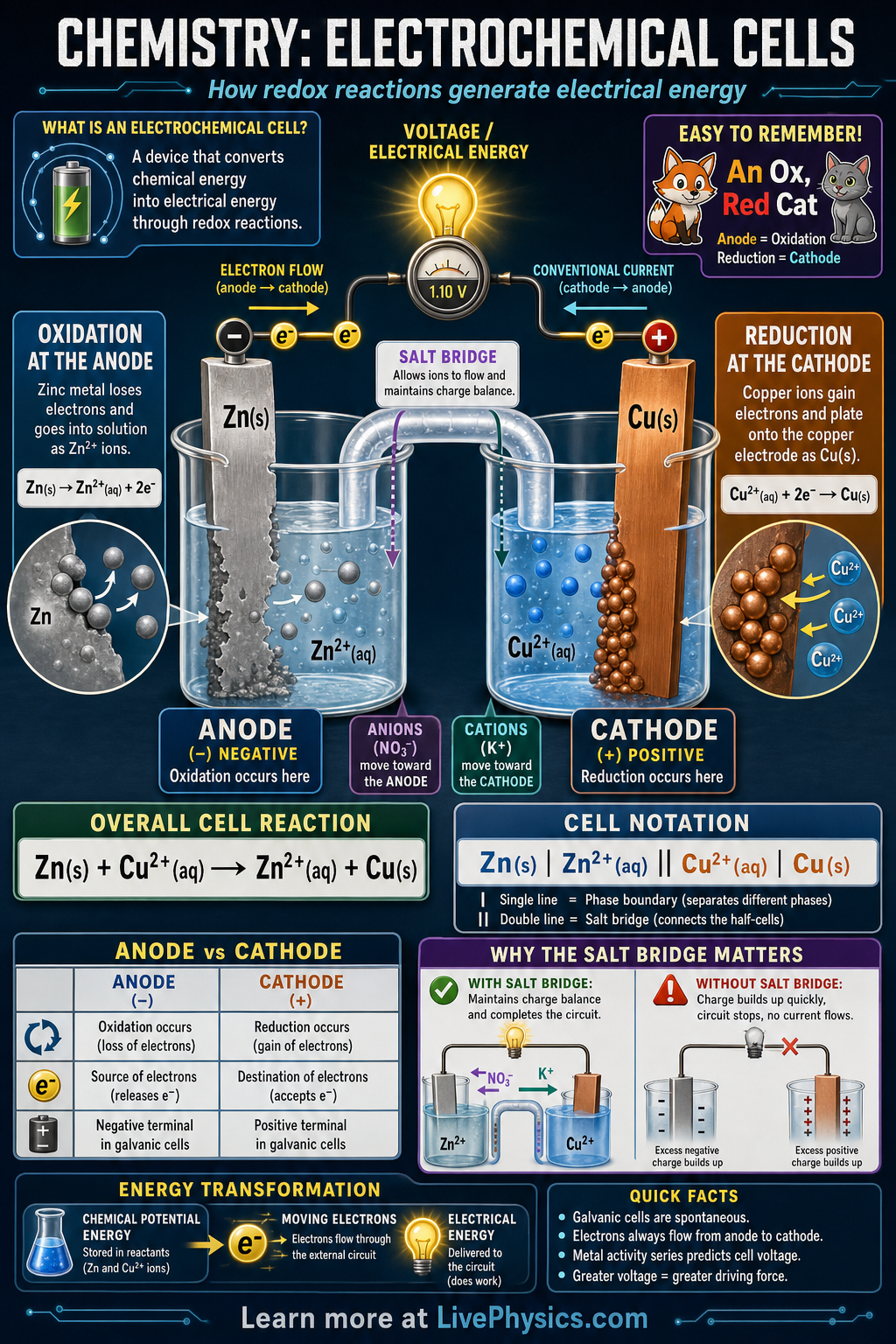
Electrochemical cells convert chemical energy into electrical energy or use electrical energy to drive chemical change. They matter because batteries, corrosion, metal plating, and many sensors all depend on electron transfer between substances. In a galvanic cell, a spontaneous redox reaction produces a voltage that can power an external circuit. Understanding the parts of the cell helps explain how charge moves through both wires and solutions.
A typical galvanic cell has two half-cells connected by a wire and a salt bridge. Oxidation occurs at the anode, where electrons are produced, and reduction occurs at the cathode, where electrons are consumed. Electrons flow through the external wire from anode to cathode, while ions move through the salt bridge to keep both solutions electrically neutral. The cell voltage depends on the reduction potentials of the two half-reactions and tells how strongly the reaction tends to occur.
Key Facts
- Oxidation is loss of electrons, and reduction is gain of electrons.
- In a galvanic cell, the anode is negative and the cathode is positive.
- Electrons flow through the external wire from anode to cathode.
- Ecell = Ecathode - Eanode using standard reduction potentials.
- A positive Ecell means the redox reaction is spontaneous under the stated conditions.
- The salt bridge allows ion flow and prevents charge buildup in the half-cells.
Vocabulary
- Electrochemical cell
- A device that connects oxidation and reduction half-reactions to transfer electrons and energy.
- Anode
- The electrode where oxidation occurs and electrons are produced.
- Cathode
- The electrode where reduction occurs and electrons are consumed.
- Salt bridge
- A connection containing ions that completes the circuit by allowing charge balance between half-cells.
- Cell potential
- The voltage produced by an electrochemical cell due to the difference in reduction tendency between the two electrodes.
Common Mistakes to Avoid
- Calling the cathode the place where oxidation happens is wrong because reduction always occurs at the cathode in both galvanic and electrolytic cells.
- Reversing electron flow is wrong because electrons leave the anode and travel through the wire to the cathode in a galvanic cell.
- Adding the two reduction potentials without changing signs correctly is wrong because Ecell = Ecathode - Eanode when using standard reduction potentials.
- Forgetting the salt bridge is wrong because the cell quickly stops if charge buildup prevents further electron flow.
Practice Questions
- 1 A galvanic cell has Zn as the anode with Ered = -0.76 V and Cu as the cathode with Ered = +0.34 V. Calculate Ecell.
- 2 A cell reaction transfers 2 mol of electrons and has Ecell = 1.10 V. Using ΔG = -nFE with F = 96485 C/mol, calculate ΔG in kJ/mol reaction.
- 3 A student removes the salt bridge from a working galvanic cell. Explain what happens to the voltage and why the cell cannot keep operating.