Chemistry: Electrochemistry: Galvanic and Electrolytic Cells
Comparing spontaneous and nonspontaneous redox reactions in cells
Comparing spontaneous and nonspontaneous redox reactions in cells
Chemistry - Grade 9-12
- 1

A galvanic cell is made from Zn and Cu electrodes. The half-reactions are Zn2+ + 2e- -> Zn with E° = -0.76 V and Cu2+ + 2e- -> Cu with E° = +0.34 V. Identify the anode, the cathode, and the direction of electron flow.
- 2
Using the Zn and Cu standard reduction potentials, calculate the standard cell potential for the cell Zn(s) | Zn2+(aq) || Cu2+(aq) | Cu(s).
- 3
Write the balanced overall reaction for the galvanic cell Zn(s) | Zn2+(aq) || Cu2+(aq) | Cu(s).
- 4
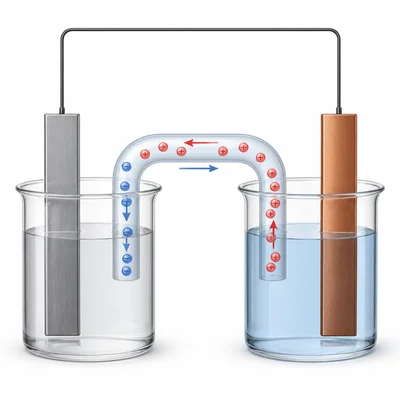
Explain the purpose of the salt bridge in a galvanic cell.
- 5

A student builds a cell with Fe and Ag electrodes. The standard reduction potentials are Fe2+ + 2e- -> Fe with E° = -0.44 V and Ag+ + e- -> Ag with E° = +0.80 V. Identify which substance is oxidized and which ion is reduced.
- 6
For the Fe and Ag cell in problem 5, write the balanced overall reaction and calculate E°cell.
- 7
Classify each cell as galvanic or electrolytic: Cell A produces electrical energy from a spontaneous redox reaction. Cell B uses a battery to force a nonspontaneous redox reaction.
- 8
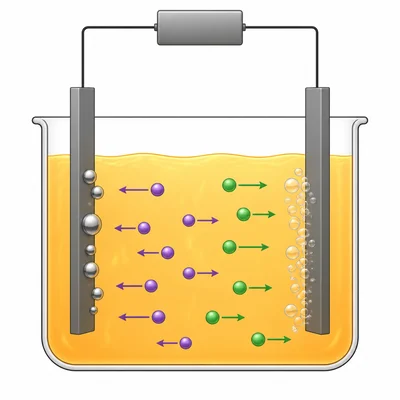
In an electrolytic cell containing molten sodium chloride, Na+ ions move toward one electrode and Cl- ions move toward the other. Identify what happens at the cathode and at the anode.
- 9
Compare the signs of the electrodes in galvanic and electrolytic cells.
- 10
A current of 2.00 A passes through a copper(II) sulfate solution for 20.0 minutes. Copper is plated according to Cu2+ + 2e- -> Cu. Calculate the mass of copper plated. Use F = 96,485 C/mol e- and molar mass of Cu = 63.55 g/mol.
- 11
A current of 5.00 A runs through molten MgCl2 for 30.0 minutes. Magnesium forms according to Mg2+ + 2e- -> Mg. Calculate the mass of magnesium produced. Use F = 96,485 C/mol e- and molar mass of Mg = 24.31 g/mol.
- 12
A cell has E°cell = -0.50 V as written. Explain whether the reaction is spontaneous as written and describe one way to make it occur in an electrolytic cell.
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems