Science: Redox Reactions and Electrochemistry
Oxidation, reduction, and electron flow in chemical systems
Oxidation, reduction, and electron flow in chemical systems
Chemistry - Grade 9-12
- 1
Define oxidation and reduction in terms of electron transfer.
- 2
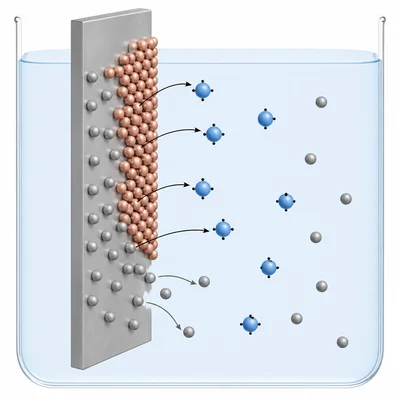
In the reaction Zn + Cu2+ -> Zn2+ + Cu, identify which species is oxidized and which species is reduced.
- 3
Assign the oxidation number of each element in H2O.
- 4
Assign the oxidation number of sulfur in SO4^2-.
- 5
Determine whether the following reaction is a redox reaction: 2Mg + O2 -> 2MgO. Explain your answer.
- 6
Write the oxidation half-reaction for magnesium in the reaction Mg -> Mg2+ + 2e-.
- 7
Write the reduction half-reaction for chlorine in the reaction Cl2 + 2e- -> 2Cl-.
- 8
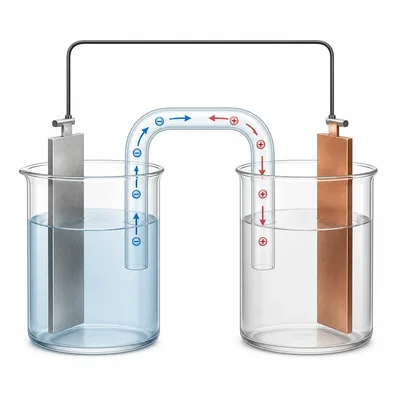
In a galvanic or voltaic cell, what is the function of the salt bridge?
- 9
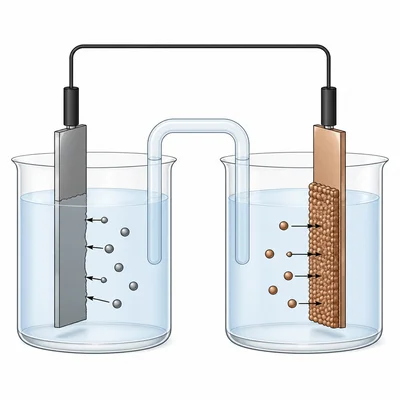
In an electrochemical cell, where does oxidation occur and where does reduction occur?
- 10

Describe the direction of electron flow in a galvanic cell.
- 11
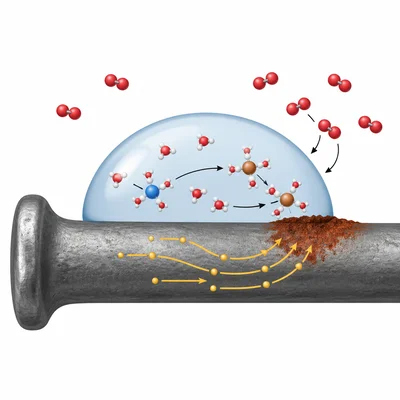
Explain why rusting of iron is considered a redox process.
- 12
A student says that the reducing agent in a reaction is the substance that gets reduced. Correct this statement and explain the role of a reducing agent.
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems