Phase Changes
Phase Changes
Related Tools
Related Labs
Related Worksheets
Related Cheat Sheets
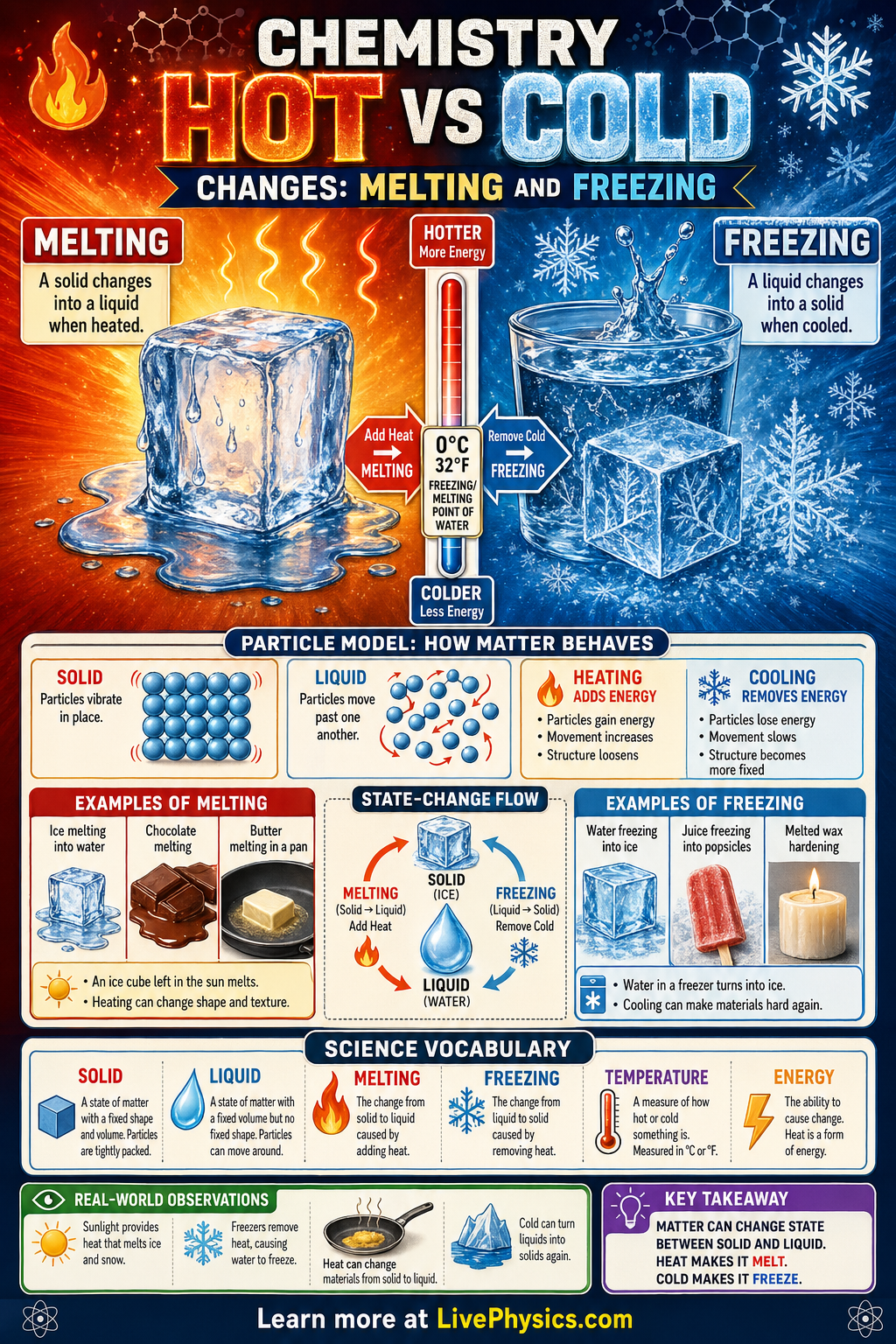
Melting and freezing are physical changes that describe how matter moves between the solid and liquid states. These changes happen when thermal energy is added or removed from a substance. Water is the most familiar example, since ice melts into liquid water when warmed and liquid water freezes into ice when cooled. Understanding these changes helps explain weather, cooking, refrigeration, and many natural cycles on Earth.
At the particle level, melting happens when particles gain enough energy to move past one another instead of staying locked in place. Freezing happens when particles lose energy and slow down until they form a more fixed arrangement. For pure water at normal atmospheric pressure, both changes occur at 0 degrees C. During melting or freezing, the temperature stays constant until the phase change is complete because energy is used to change state rather than raise or lower temperature.
Key Facts
- Melting is the change from solid to liquid.
- Freezing is the change from liquid to solid.
- For pure water at atm, melting point = freezing point = degrees C.
- for melting or freezing, where is heat, is mass, and is latent heat of fusion.
- If heat is added, particles gain kinetic energy and melting becomes more likely.
- During a phase change, temperature remains constant until all of the substance has changed state.
Vocabulary
- phase change
- A phase change is a change of state, such as from solid to liquid or liquid to solid.
- melting point
- The melting point is the temperature at which a solid changes into a liquid.
- freezing point
- The freezing point is the temperature at which a liquid changes into a solid.
- thermal energy
- Thermal energy is the energy associated with the motion of particles in a substance.
- latent heat of fusion
- Latent heat of fusion is the energy needed per unit mass to melt a solid or released per unit mass when a liquid freezes.
Common Mistakes to Avoid
- Thinking melting and dissolving are the same thing, which is wrong because melting is a change of state while dissolving involves mixing one substance into another.
- Assuming temperature always changes when heat is added or removed, which is wrong because during melting or freezing the energy goes into changing the state instead.
- Believing water freezes at any cold temperature, which is wrong because pure water at normal pressure freezes specifically at 0 degrees C.
- Confusing particle size with particle motion, which is wrong because particles do not get bigger or smaller during melting or freezing, they only change how they move and arrange themselves.
Practice Questions
- 1 A block of ice melts completely. If the latent heat of fusion of water is , how much heat is required?
- 2 How much heat is released when of liquid water at freezes? Use .
- 3 A cup of ice water stays at 0 degrees C while some of the ice is still present. Explain why the temperature does not rise even though heat is entering the cup.