Chemistry: Thermochemistry: Enthalpy and Hess's Law
Calculating heat, enthalpy changes, and reaction pathways
Calculating heat, enthalpy changes, and reaction pathways
Chemistry - Grade 9-12
- 1
The combustion of methane is represented by CH4(g) + 2O2(g) -> CO2(g) + 2H2O(l), delta H = -890 kJ per mole of CH4. How much heat is released when 2.50 mol of CH4 burns completely?
- 2
A reaction warms 150.0 g of water from 22.0°C to 31.5°C. Use q = mc delta T and c = 4.184 J/g°C to find the heat gained by the water. Then state the heat change for the reaction.
- 3

When ammonium nitrate dissolves in water, the temperature of the solution decreases. State whether the process is endothermic or exothermic and explain the sign of delta H.
- 4
Use the data to find delta H for CO(g) + 1/2O2(g) -> CO2(g). Given: C(s) + O2(g) -> CO2(g), delta H = -393.5 kJ. C(s) + 1/2O2(g) -> CO(g), delta H = -110.5 kJ.
- 5
Use standard enthalpies of formation to calculate delta H for C2H5OH(l) + 3O2(g) -> 2CO2(g) + 3H2O(l). Values: C2H5OH(l) = -277.7 kJ/mol, CO2(g) = -393.5 kJ/mol, H2O(l) = -285.8 kJ/mol, O2(g) = 0 kJ/mol.
- 6
For N2(g) + 3H2(g) -> 2NH3(g), delta H = -92.4 kJ. What is delta H for 4NH3(g) -> 2N2(g) + 6H2(g)?
- 7
Use Hess's Law to find delta H for 2SO2(g) + O2(g) -> 2SO3(g). Given: 2S(s) + 3O2(g) -> 2SO3(g), delta H = -790.4 kJ. S(s) + O2(g) -> SO2(g), delta H = -296.8 kJ.
- 8
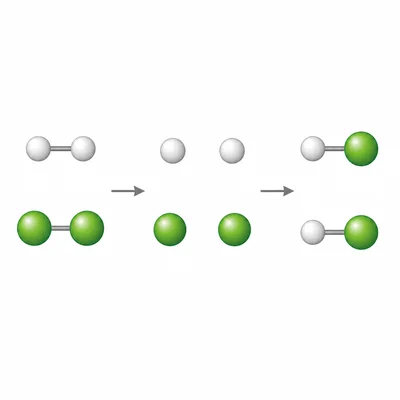
Estimate delta H for H2(g) + Cl2(g) -> 2HCl(g) using bond enthalpies. H-H = 436 kJ/mol, Cl-Cl = 243 kJ/mol, H-Cl = 431 kJ/mol.
- 9
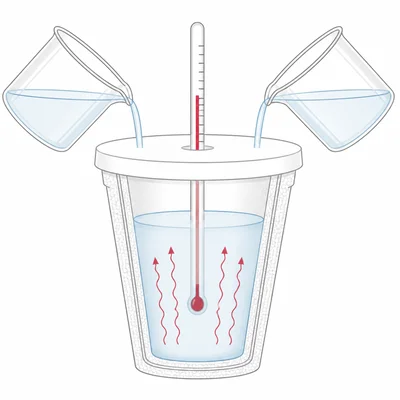
In a coffee-cup calorimeter, 50.0 mL of 1.00 M HCl reacts with 50.0 mL of 1.00 M NaOH. The temperature rises from 22.0°C to 28.7°C. Assume the solution has a mass of 100.0 g and c = 4.184 J/g°C. Calculate delta H in kJ per mole of water formed.
- 10
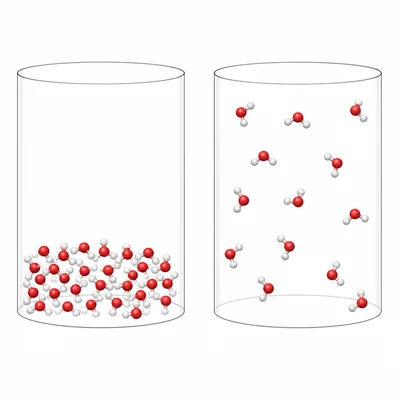
Explain why the state symbols in a thermochemical equation matter. Use H2O(l) and H2O(g) as part of your explanation.
- 11
Use standard enthalpies of formation to calculate delta H for CaCO3(s) -> CaO(s) + CO2(g). Values: CaCO3(s) = -1207.0 kJ/mol, CaO(s) = -635.1 kJ/mol, CO2(g) = -393.5 kJ/mol.
- 12
Use Hess's Law to calculate delta H for N2(g) + 2O2(g) -> 2NO2(g). Given: N2(g) + O2(g) -> 2NO(g), delta H = +180.5 kJ. 2NO(g) + O2(g) -> 2NO2(g), delta H = -114.1 kJ.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems