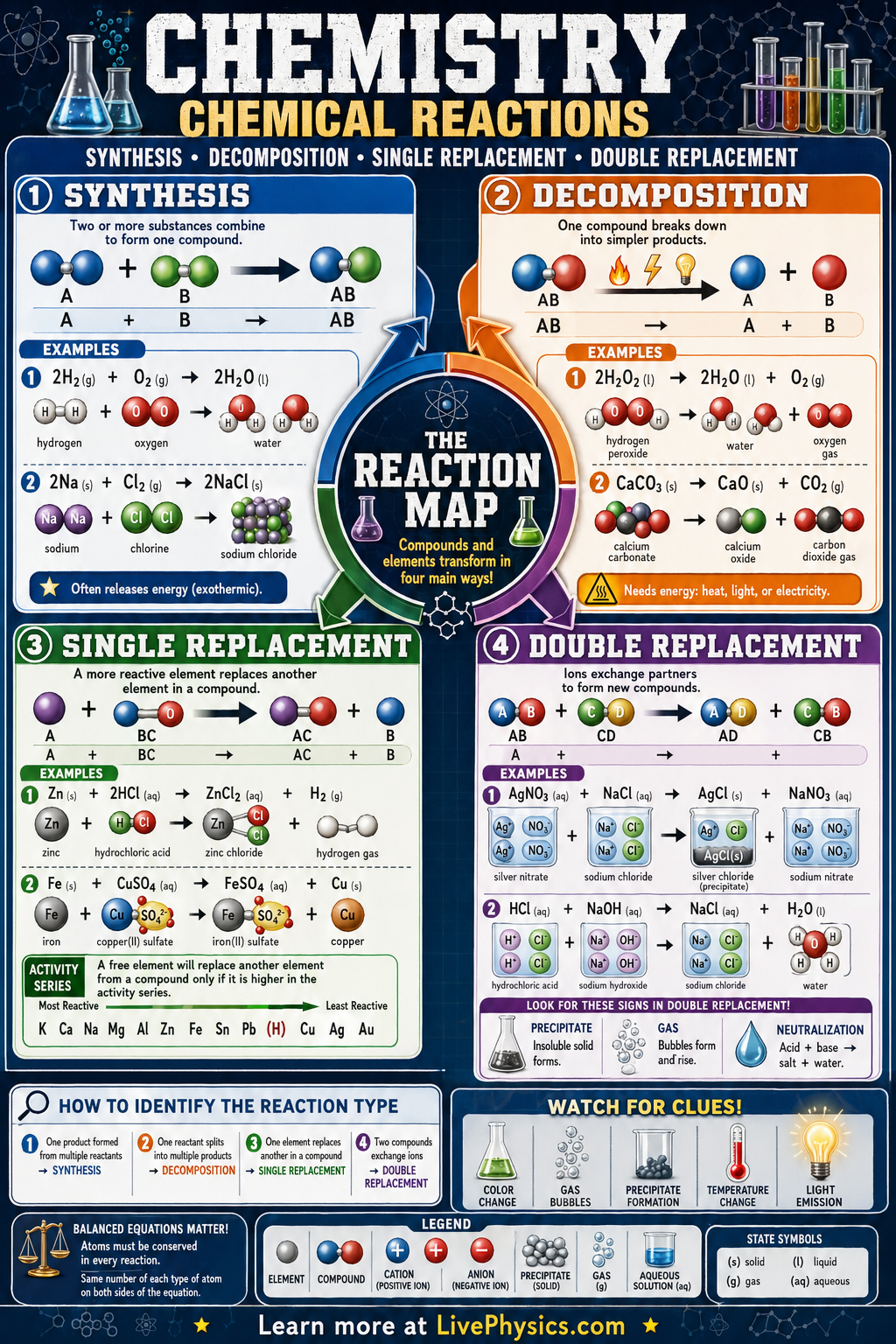
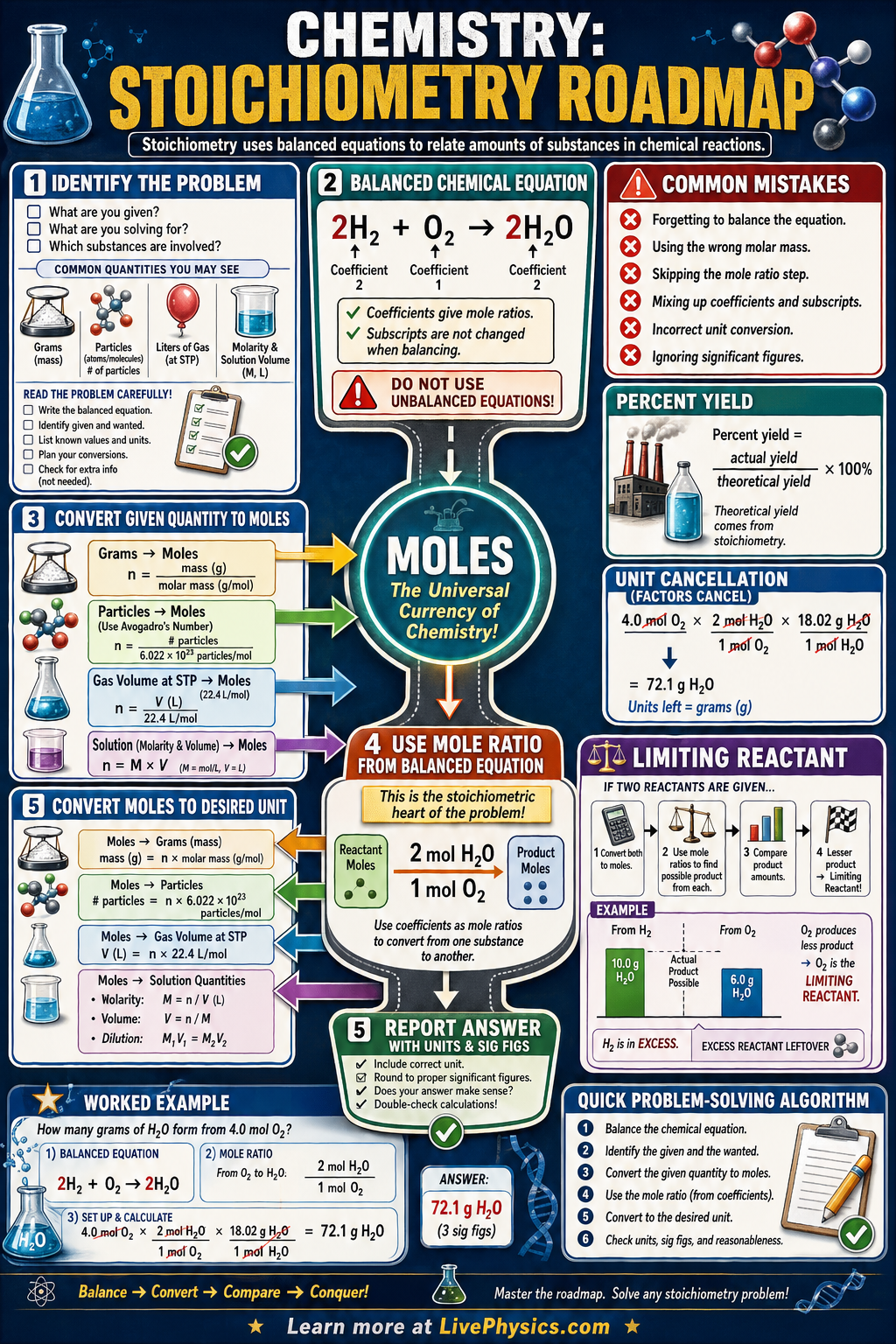
Chemical Reactions Mission
Master stoichiometry, equilibrium, acid-base chemistry, redox, and electrochemistry - essential for AP Chemistry success.
9 activities · Work through in order for a complete lesson arc
Molar Mass & Stoichiometry Calculator
Enter any chemical formula to compute molar mass, element breakdown with mass percentages, and step-by-step calculations. Supports parenthesized groups.
pH, pOH & Acid-Base Calculator
Enter pH, pOH, [H+], or [OH-] to compute all four. Color-coded pH scale bar, acid/base classification, and step-by-step KaTeX.
ICE / Q / K / Ksp Equilibrium Solver
Four equilibrium chemistry modes: build ICE tables and solve for equilibrium concentrations, compare reaction quotient Q to K and predict shift direction, calculate molar solubility from Ksp with optional common ion effect, and solve weak acid/base Ka or Kb for pH and percent ionization. Step-by-step solutions with KaTeX.
ICE tables for equilibrium, Q vs K, and Ksp.
Oxidation-Reduction (Redox) Calculator
Enter a chemical reaction to assign oxidation numbers, identify what is oxidized and reduced, and find oxidizing and reducing agents with electron transfer count.
Buffer & Titration Curve Explorer
Interactive titration curve tool for strong acid-strong base, weak acid-strong base, weak base-strong acid, and diprotic acid titrations. See real-time pH curve, buffer regions, Henderson-Hasselbalch equation, equivalence points, buffer capacity, and indicator color overlays.
Chemical Kinetics Lab
Simulate reaction rates with the Arrhenius equation. Adjust concentration, temperature, and catalysts to observe how particle collision frequency and rate constants change
Titration Lab
Simulate acid-base titration with a virtual burette. Watch pH curves develop in real time, find equivalence points, and compare strong and weak acid behavior with indicator color changes
Calorimetry Lab
Mix substances at different temperatures and observe heat exchange over time. Calculate specific heat, compare coffee cup and bomb calorimeters, and model heat loss
Electrochemistry Lab
Build galvanic and electrolytic cells by selecting anode and cathode metals. Calculate cell potential with the Nernst equation and explore Gibbs free energy relationships
Capstone - galvanic cells and electrolysis.
Study Materials
Worksheets