Chemistry: Quantum Mechanical Model of the Atom
Orbitals, quantum numbers, and electron configurations
Orbitals, quantum numbers, and electron configurations
Chemistry - Grade 9-12
- 1
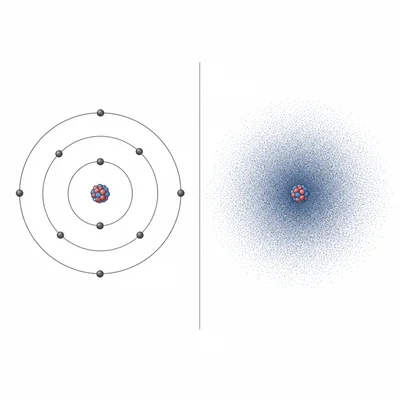
Describe how the quantum mechanical model of the atom is different from the Bohr model of the atom.
- 2
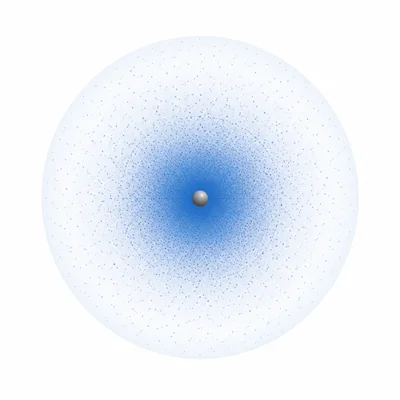
In the quantum mechanical model, what is an orbital?
- 3
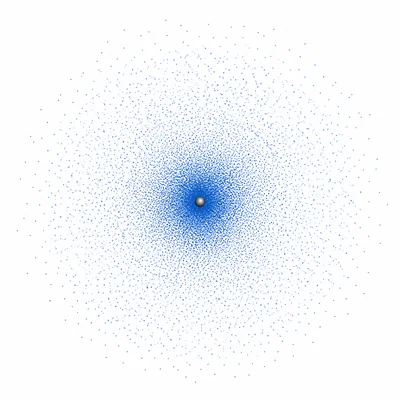
Explain why the word probability is important when describing the location of an electron.
- 4
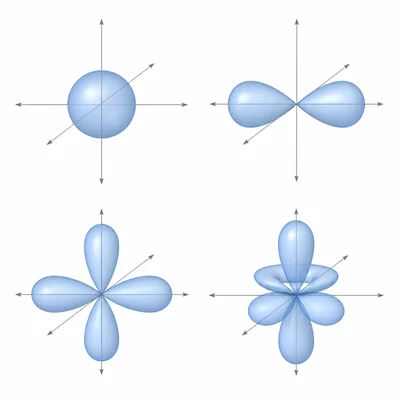
Identify the four types of atomic orbitals commonly used in high school chemistry and give the maximum number of electrons each sublevel can hold.
- 5
A student says, "An electron in a 3p orbital is in the third main energy level." Explain whether the student is correct.
- 6
For the orbital label 4d, identify the principal energy level and the sublevel.
- 7
Complete the electron configuration for carbon, which has 6 electrons.
- 8
Write the electron configuration for sodium, which has 11 electrons.
- 9
Use the Aufbau principle to explain why the 4s sublevel is filled before the 3d sublevel in many electron configurations.
- 10
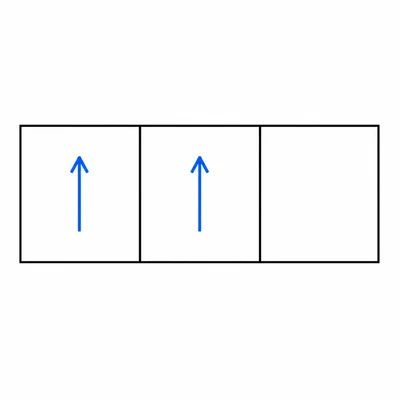
State Hund's rule and apply it to the three 2p orbitals when carbon has two electrons in 2p.
- 11
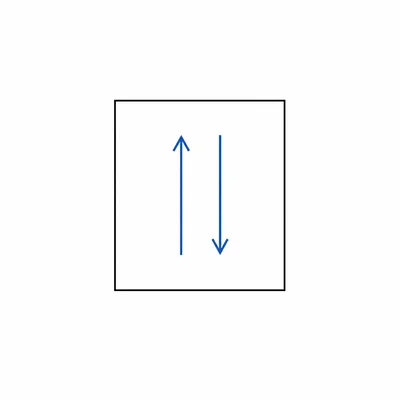
State the Pauli exclusion principle and explain what it means for one orbital.
- 12
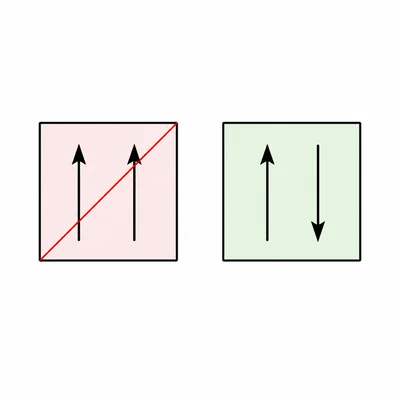
An orbital box diagram shows one box with two arrows both pointing up. Explain what is wrong with this diagram.
- 13
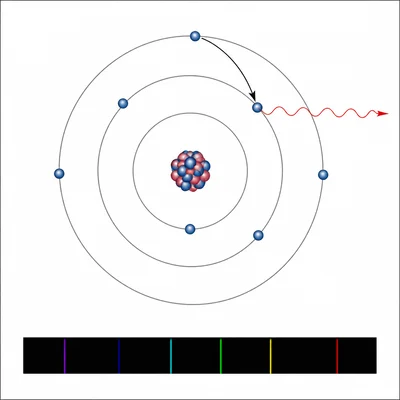
Explain how an atomic emission spectrum provides evidence that electrons have quantized energy levels.
- 14
A photon has an energy of 3.20 x 10^-19 joules. Use E = hν, with h = 6.63 x 10^-34 J·s, to calculate the frequency of the photon.
- 15
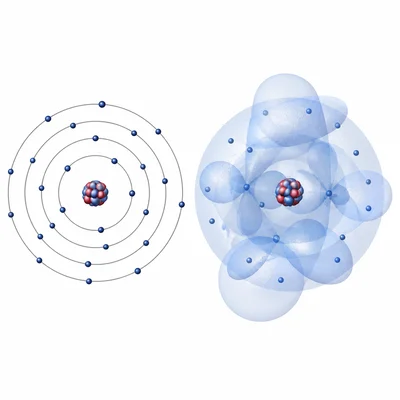
Explain why the quantum mechanical model is more useful than the Bohr model for describing atoms with many electrons.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems