Mixtures vs Pure Substances
Mixtures and Pure Substances
Related Tools
Related Labs
Related Worksheets
Related Cheat Sheets
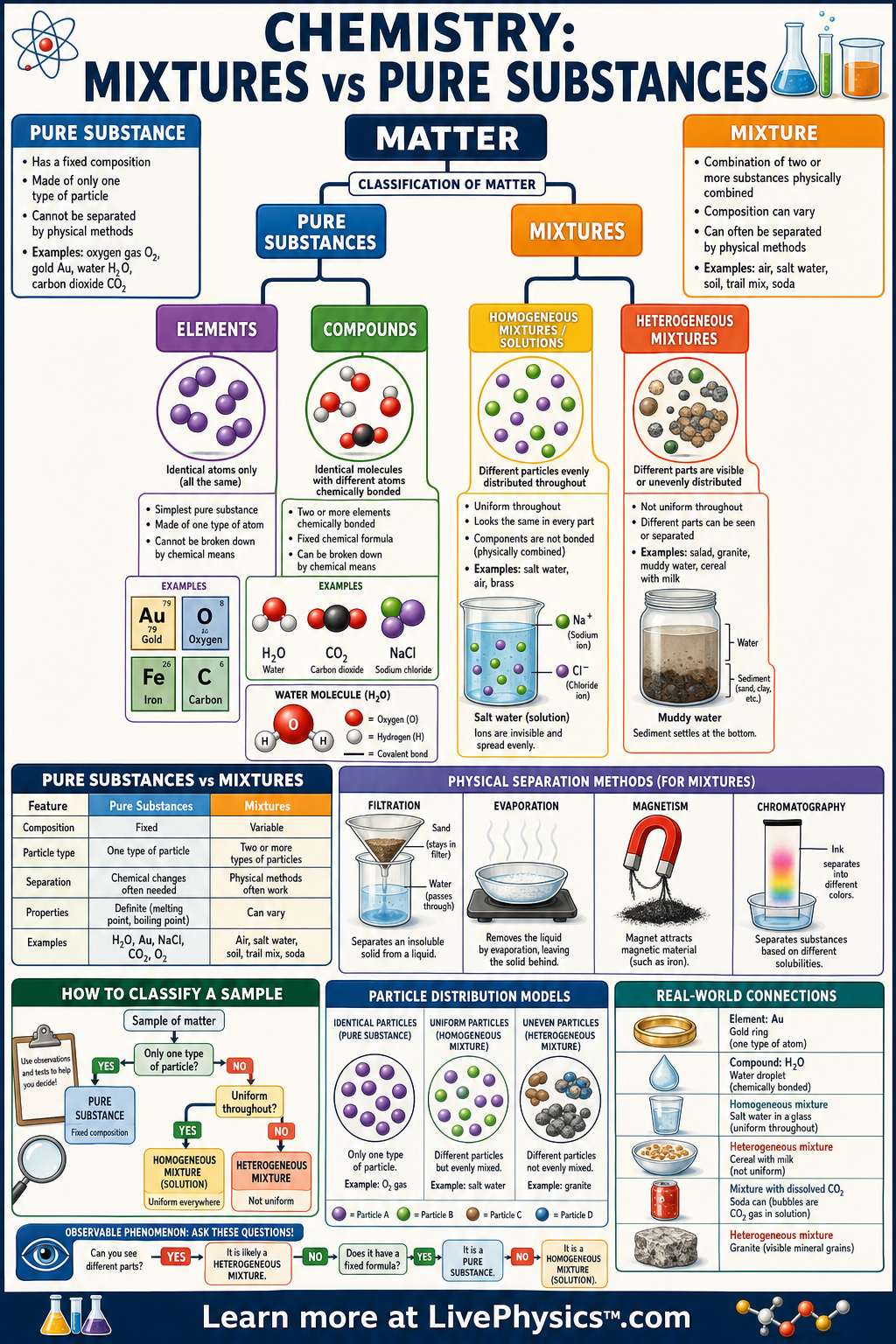
Matter can be classified by what it is made of and whether its parts can be separated by physical means. A pure substance has a fixed composition, while a mixture contains two or more substances combined without changing their chemical identities. This distinction matters because it helps scientists identify materials, choose separation methods, and understand everyday substances like air, salt water, and aluminum foil. Learning this classification gives students a clear map for organizing the many materials they see in labs and daily life.
Pure substances are either elements or compounds, and each has particles arranged in a consistent way. Mixtures can be homogeneous, meaning uniform throughout, or heterogeneous, meaning different parts can be seen or detected. Physical properties such as boiling point, density, solubility, magnetism, and particle size can be used to separate mixtures. Chemical formulas, particle diagrams, and real-world examples all help show whether a sample is a pure substance or a mixture.
Key Facts
- Matter is anything that has mass and takes up space.
- Pure substances have a fixed composition and include elements and compounds.
- Elements contain only one kind of atom, such as O2, Fe, or Cu.
- Compounds contain two or more elements chemically bonded in a fixed ratio, such as H2O or CO2.
- Mixtures contain two or more substances physically combined, so their composition can vary.
- Mass percent = mass of part / mass of whole x 100%
Vocabulary
- Pure substance
- A material with a fixed composition and only one type of element or compound throughout.
- Element
- A pure substance made of only one kind of atom.
- Compound
- A pure substance made of two or more different elements chemically bonded in a fixed ratio.
- Homogeneous mixture
- A mixture that looks uniform throughout because its parts are evenly distributed.
- Heterogeneous mixture
- A mixture with parts that are not evenly distributed and may be seen as different regions or phases.
Common Mistakes to Avoid
- Calling all clear liquids pure substances is wrong because many clear liquids, such as salt water or vinegar, are homogeneous mixtures.
- Thinking compounds are mixtures is wrong because compounds have chemically bonded atoms in fixed ratios, while mixtures are physically combined and can vary in composition.
- Classifying a substance by appearance only is wrong because some mixtures look uniform and require tests such as evaporation, chromatography, or density measurement to identify.
- Assuming mixtures always have visible parts is wrong because homogeneous mixtures like air and sugar water have particles spread evenly at a scale too small to see.
Practice Questions
- 1 A student dissolves 12 g of salt in 88 g of water. What is the mass percent of salt in the saltwater mixture?
- 2 A snack mix contains 30 g of raisins, 45 g of cereal, and 25 g of peanuts. What is the total mass of the mixture, and what percent of the mixture is peanuts?
- 3 Classify each sample as an element, compound, homogeneous mixture, or heterogeneous mixture: oxygen gas, carbon dioxide, muddy water, and lemonade with all sugar fully dissolved. Explain the reason for each classification.