Chemistry: Elements, Compounds, and Mixtures
Classifying matter by what it is made of
Classifying matter by what it is made of
Chemistry - Grade 6-8
- 1
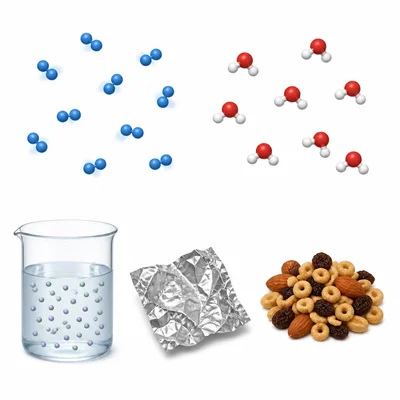
Classify each sample as an element, compound, or mixture: oxygen gas (O2), water (H2O), salt water, aluminum foil, and trail mix.
- 2
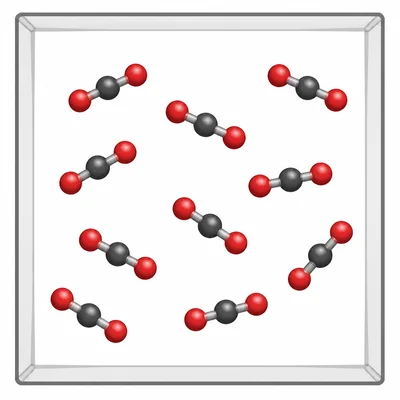
A student says, "Carbon dioxide is a mixture because it contains carbon and oxygen." Explain why this statement is incorrect.
- 3

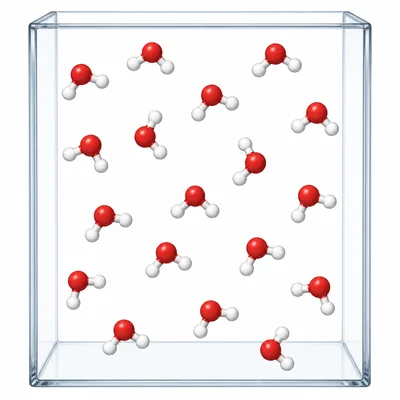
Look at a particle diagram with identical single atoms spread throughout a box. What type of matter does the diagram show, and how do you know?
- 4
Look at a particle diagram with identical molecules. Each molecule has one large red atom bonded to two small white atoms. What type of matter does the diagram show, and how do you know?
- 5

A bowl of cereal with milk contains cereal pieces, milk, and fruit slices. Is it a pure substance or a mixture? Explain your answer.
- 6
Which of the following are pure substances: gold, air, sugar (C6H12O6), vegetable soup, and helium? Explain your choices.
- 7
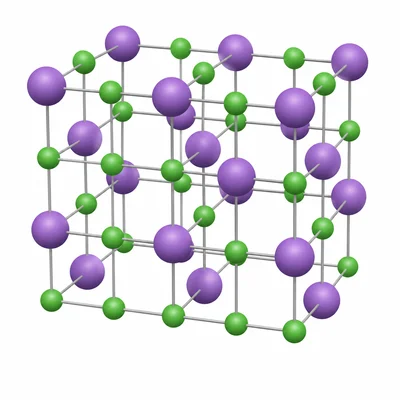
Table salt has the chemical formula NaCl. What does this formula tell you about table salt?
- 8

A sample contains iron filings mixed with sand. Describe one physical method you could use to separate the mixture.
- 9

A clear liquid is made by dissolving sugar in water. Is this a homogeneous mixture or a heterogeneous mixture? Explain your answer.
- 10

A salad contains lettuce, tomatoes, cucumbers, and cheese. Is it a homogeneous mixture or a heterogeneous mixture? Explain your answer.
- 11
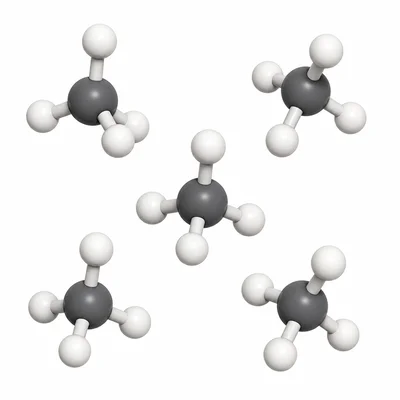
Methane has the chemical formula CH4. Is methane an element, a compound, or a mixture? Explain how you know.
- 12
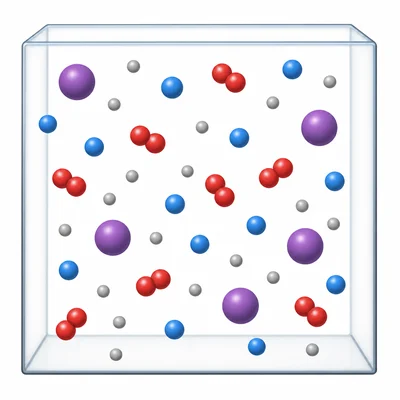
Air contains nitrogen, oxygen, carbon dioxide, argon, and other gases. Why is air classified as a mixture instead of a compound?
- 13
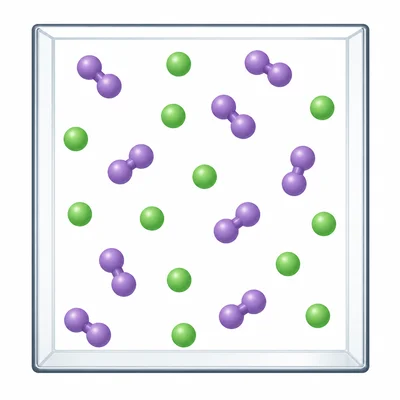
Look at a particle diagram showing two kinds of particles: some single green atoms and some pairs of purple atoms bonded together. The particles are mixed in the same box but are not bonded to each other. What type of matter is shown?
- 14
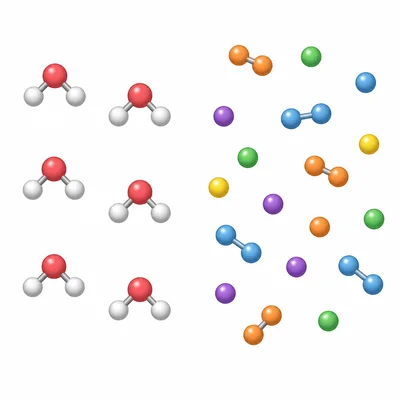
Explain the difference between a compound and a mixture using the words "chemically bonded" and "physically combined."
- 15

A teacher gives you three unknown samples. Sample A has only one type of atom. Sample B has two types of atoms bonded in identical molecules. Sample C has several substances that can be separated by filtering. Classify Sample A, Sample B, and Sample C.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems