Carbon, Nitrogen, and Phosphorus Cycles
Carbon, Nitrogen, and Phosphorus Cycles
Related Tools
Related Labs
Related Worksheets
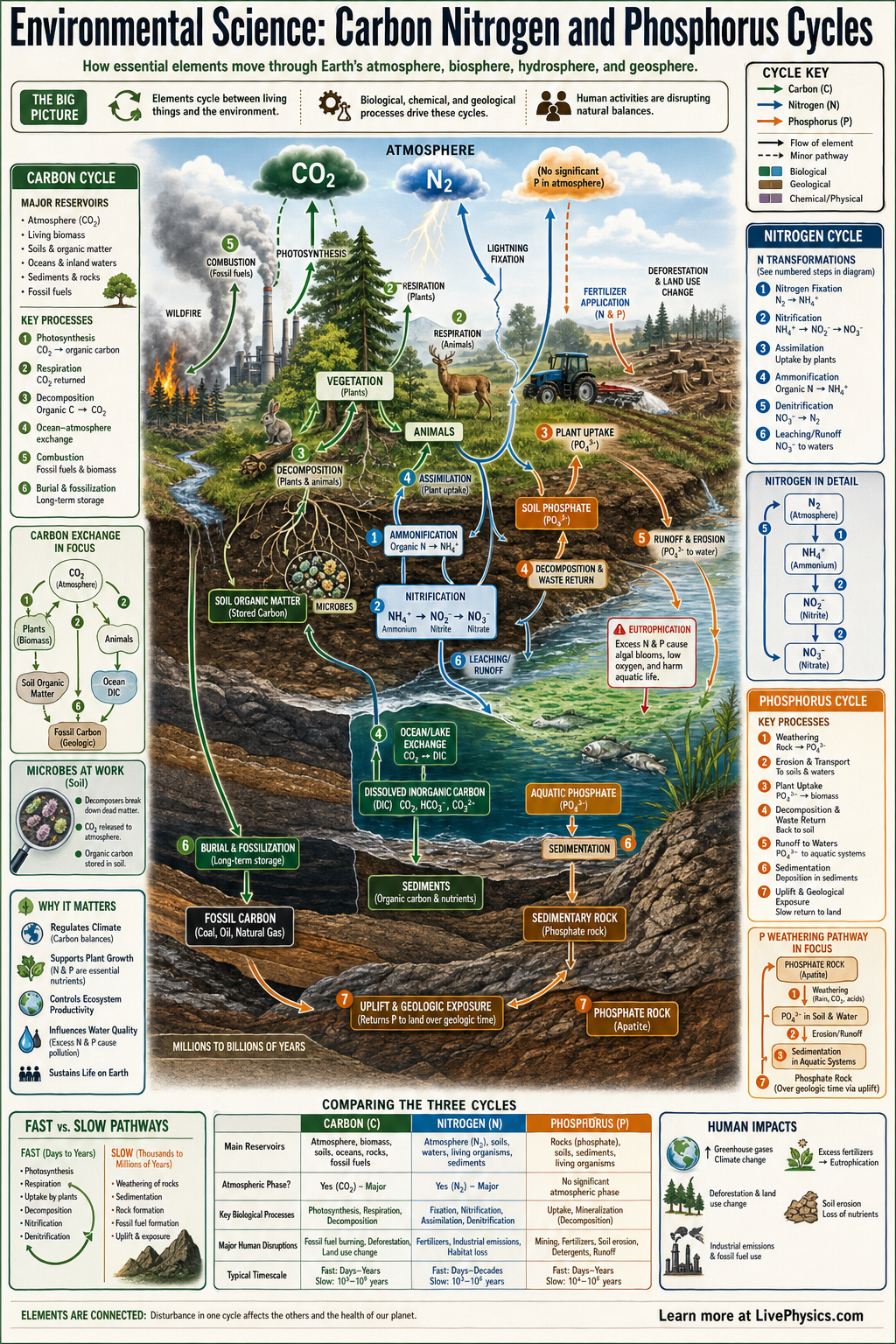
The carbon, nitrogen, and phosphorus cycles describe how key elements move through air, water, soil, rocks, and living things. These cycles matter because all organisms need these elements to build cells, store energy, and carry out life processes. Ecosystems stay productive only when these materials are continuously recycled. Human activities can speed up, slow down, or redirect these flows and cause major environmental change.
Carbon moves strongly through the atmosphere and living biomass, nitrogen depends heavily on microbes to change it into usable forms, and phosphorus mostly cycles through rocks, soil, and water because it has no major gaseous phase. Plants take in carbon dioxide, nitrate, ammonium, and phosphate, then animals obtain these elements by feeding. Decomposition returns them to soil, water, and air. Farming, fossil fuel burning, mining, and fertilizer runoff can disrupt all three cycles and lead to climate change, eutrophication, and soil degradation.
Key Facts
- Photosynthesis removes carbon dioxide from air: 6CO2 + 6H2O -> C6H12O6 + 6O2
- Cellular respiration returns carbon to the atmosphere: C6H12O6 + 6O2 -> 6CO2 + 6H2O + energy
- Nitrogen fixation converts atmospheric nitrogen into ammonia: N2 -> NH3
- Nitrification changes ammonium into nitrate in steps: NH4+ -> NO2- -> NO3-
- Plants commonly absorb nitrogen as NO3- or NH4+ and phosphorus as PO4^3-
- Phosphorus often enters ecosystems by weathering of phosphate rock and can leave soils by runoff into water
Vocabulary
- Photosynthesis
- The process by which plants, algae, and some microbes use light energy to turn carbon dioxide and water into sugars.
- Nitrogen fixation
- The conversion of atmospheric nitrogen gas into ammonia or related compounds that living things can use.
- Nitrification
- A bacterial process that converts ammonium first into nitrite and then into nitrate.
- Weathering
- The physical and chemical breakdown of rocks that releases minerals such as phosphate into soil and water.
- Eutrophication
- The over-enrichment of water with nutrients, often causing algal blooms and low oxygen levels.
Common Mistakes to Avoid
- Thinking phosphorus has a major atmospheric cycle, which is wrong because phosphorus usually moves through rocks, soil, water, and organisms rather than as a common gas in air.
- Assuming plants can use atmospheric N2 directly, which is wrong because most plants need nitrogen first converted by microbes into ammonium or nitrate.
- Confusing photosynthesis with respiration, which is wrong because photosynthesis stores carbon in sugars while respiration releases carbon dioxide back to the environment.
- Believing fertilizer always stays in the field, which is wrong because nitrogen and phosphorus can leach or run off into waterways and cause pollution.
Practice Questions
- 1 A forest removes 1200 kg of carbon from the atmosphere by photosynthesis in one season, but respiration and decomposition return 850 kg. What is the net carbon storage change for that season?
- 2 A soil sample contains 18 mg/L NH4+ that is fully converted by nitrification. If each mole of NH4+ becomes one mole of NO3-, how many mg/L of nitrogen are present in nitrate form after the conversion, assuming no losses?
- 3 Explain why adding large amounts of phosphate fertilizer to farmland can increase algal blooms in a nearby lake even though phosphorus is essential for plant growth.