Chemistry: Nuclear Chemistry: Fission, Fusion, and Decay
Balancing nuclear reactions and understanding radioactive processes
Balancing nuclear reactions and understanding radioactive processes
Chemistry - Grade 9-12
- 1
In alpha decay, uranium-238 becomes thorium-234 and releases an alpha particle. Write the balanced nuclear equation using isotope notation.
- 2
Carbon-14 undergoes beta-minus decay to form nitrogen-14. Write the balanced nuclear equation and explain what happens to the atomic number.
- 3
A radioactive isotope has a half-life of 8 days. If a sample starts with 80 grams, how many grams remain after 24 days?
- 4
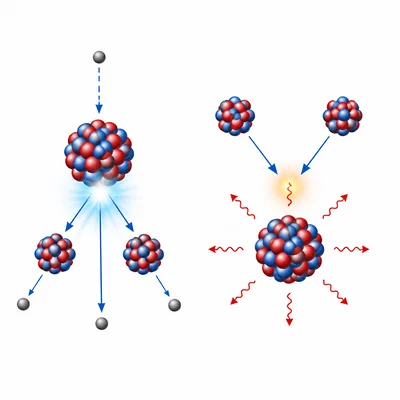
Compare nuclear fission and nuclear fusion in terms of what happens to atomic nuclei.
- 5
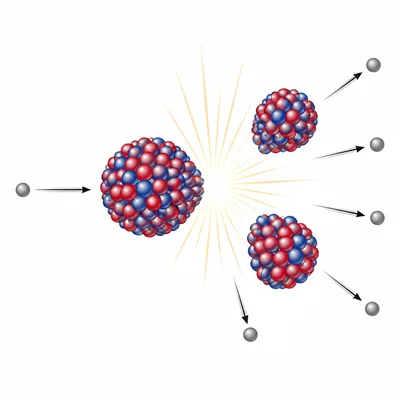
Complete and balance this nuclear equation: U-235 + n-1 -> Ba-141 + Kr-92 + ___ n-1.
- 6
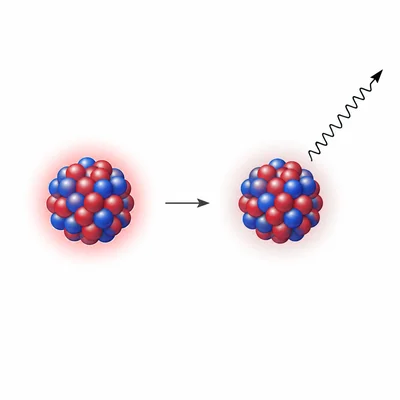
A nucleus emits a gamma ray. Describe how the mass number and atomic number of the nucleus change.
- 7
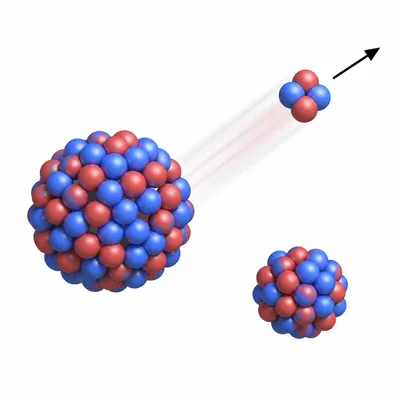
Radium-226 decays by alpha emission. Identify the daughter isotope by mass number and element name.
- 8
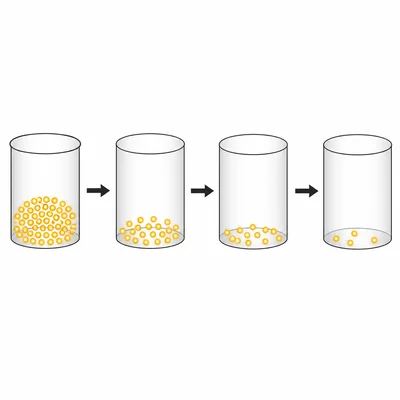
A 200 gram sample of iodine-131 decays to 25 grams in 24 days. How many half-lives have passed, and what is the half-life of iodine-131 in this situation?
- 9
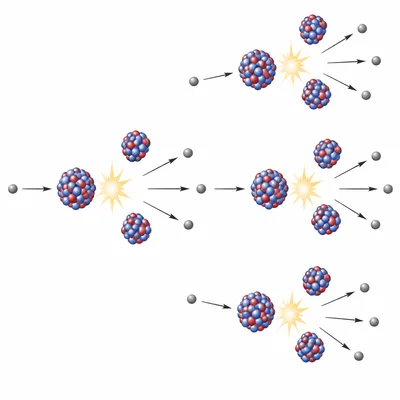
Explain why a nuclear chain reaction can occur during fission of uranium-235.
- 10
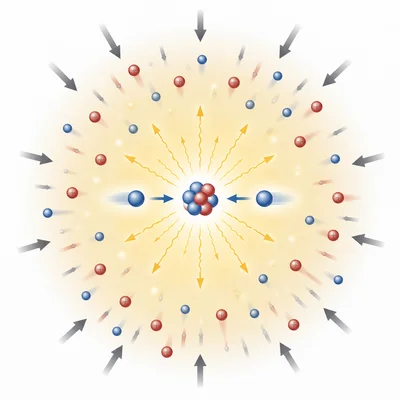
Hydrogen isotopes can fuse in stars to form helium. Explain why fusion requires extremely high temperature and pressure.
- 11
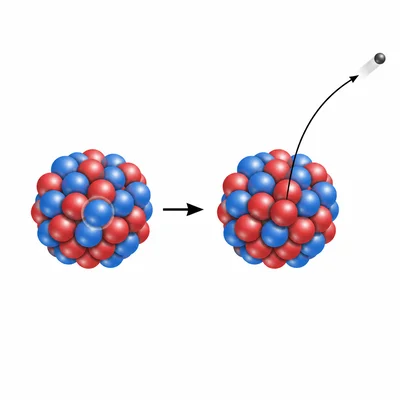
Identify the type of decay represented by this change: P-32 -> S-32 + beta particle.
- 12
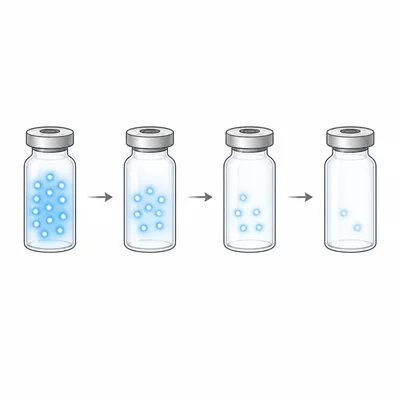
A medical tracer has a half-life of 6 hours. A patient receives a dose with an activity of 64 units. How much activity remains after 18 hours?
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems