Chemistry: Solution Chemistry: Molarity and Dilution
Calculating concentration, moles, volume, and dilution
Calculating concentration, moles, volume, and dilution
Chemistry - Grade 9-12
- 1
A student dissolves 0.50 mol of NaCl in enough water to make 2.0 L of solution. What is the molarity of the NaCl solution?
- 2
A solution is made by dissolving 20.2 g of KNO3 in enough water to make 500.0 mL of solution. The molar mass of KNO3 is 101.1 g/mol. What is the molarity?
- 3
How many grams of NaCl are needed to prepare 250.0 mL of a 0.150 M NaCl solution? The molar mass of NaCl is 58.44 g/mol.
- 4
A lab needs 500.0 mL of 0.250 M HCl made from a 2.00 M HCl stock solution. What volume of the stock solution is needed?
- 5
A student dilutes 100.0 mL of 0.750 M CuSO4 solution to a final volume of 600.0 mL. What is the final molarity?
- 6
A 50.0 mL sample of 0.200 M NaOH is diluted by adding water until the final volume is 200.0 mL. What is the new concentration of NaOH?
- 7
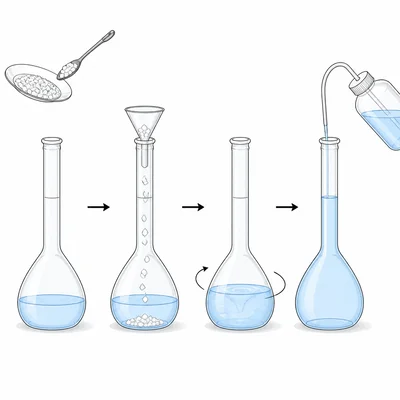
Explain why a chemist who prepares a solution from a solid should dissolve the solid in some water first, then add water until the solution reaches the final volume mark.
- 8
Which solution contains more moles of solute: 0.300 L of 0.500 M glucose or 0.200 L of 0.800 M glucose? Show your work.
- 9
A stock solution has a concentration of 6.00 M. A student transfers 25.0 mL of this stock solution into a 250.0 mL volumetric flask and fills to the mark with water. What is the concentration of the diluted solution?
- 10
A solution contains 0.0750 mol of CaCl2 in 125 mL of solution. What is the molarity of CaCl2?
- 11
How many milliliters of a 0.400 M Na2CO3 solution contain 0.0200 mol of Na2CO3?
- 12
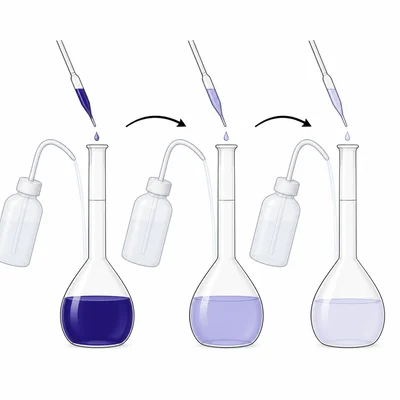
A student starts with 1.00 M food dye solution. The student makes a 1:10 dilution by placing 10.0 mL of the dye into a 100.0 mL volumetric flask and filling to the mark. Then the student repeats the same 1:10 dilution using the first diluted solution. What is the final concentration after the second dilution?
- 13
A 2.50 M solution is diluted to a final volume of 1.00 L. If 125 mL of the stock solution was used, what is the final concentration?
- 14
A student needs to prepare 100.0 mL of 0.250 M MgCl2 solution from solid MgCl2. The molar mass of MgCl2 is 95.21 g/mol. What mass of MgCl2 should the student measure?
- 15
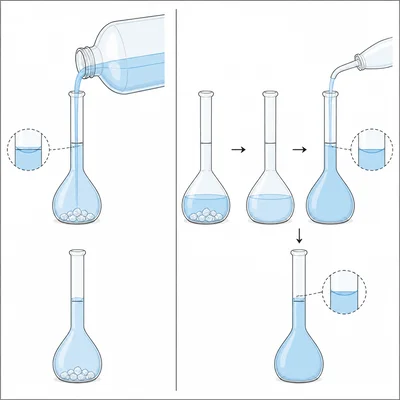
A student writes, 'To make 1.00 L of 0.100 M solution, add 1.00 L of water to 0.100 mol of solute.' Explain what is wrong with this statement and write a corrected version.
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems