Chemistry: Acids, Bases, and the pH Scale
Classifying substances and interpreting pH values
Classifying substances and interpreting pH values
Chemistry - Grade 6-8
- 1
A sample has a pH of 3. Is it acidic, basic, or neutral? Explain how you know.
- 2
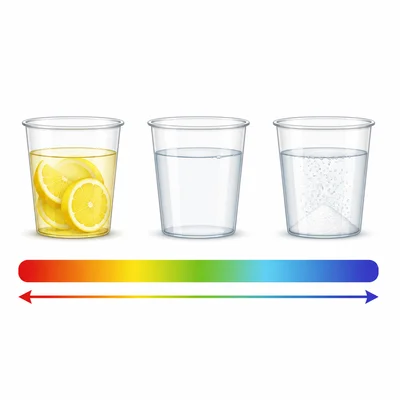
A student tests three liquids. Lemon juice has pH 2, pure water has pH 7, and baking soda solution has pH 9. List the liquids from most acidic to most basic.
- 3

What pH value is considered neutral? Name one common substance that is close to neutral.
- 4
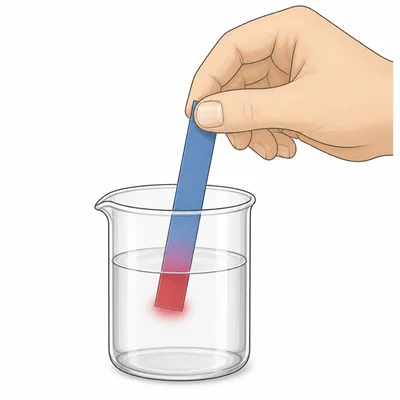
Blue litmus paper turns red when dipped into an unknown liquid. What type of substance is the liquid most likely to be?
- 5

Red litmus paper stays red when dipped into vinegar. Does this result prove that vinegar is neutral? Explain.
- 6

A cleaning solution has a pH of 12. Describe one safety precaution you should use when handling it and explain why.
- 7
A student says, "All acids are dangerous." Explain why this statement is too simple.
- 8
Two solutions have pH values of 4 and 6. Which solution is more acidic, and how many times more acidic is it based on the pH scale?
- 9

A pH indicator turns green in a liquid. The indicator chart says green means pH 7. What can you conclude about the liquid?
- 10

An acid and a base are mixed carefully in the correct amounts, and the final solution has a pH of 7. What type of reaction happened?
- 11

Classify each substance as acid, base, or neutral: stomach acid pH 1, soap solution pH 10, and salt water pH 7.
- 12

A fish tank should stay near pH 7. A test shows the water is pH 5. Is the water too acidic or too basic, and what should the owner do before adding any chemical treatment?
- 13

The table shows four unknown liquids: A has pH 8, B has pH 2, C has pH 7, and D has pH 13. Which liquid is the strongest base?
- 14

Explain the difference between a strong acid and a weak acid in middle school terms.
- 15

A student measures orange juice at pH 3 and milk at pH 6. Which is closer to neutral, and how do you know?
Related Tools
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 6-8 Worksheets
Ratios & Proportions
Math · 8 problems
Forces & Motion
Physics · 8 problems
Figurative Language
Language Arts · 8 problems
US Government & Civics
Social Studies · 8 problems