Chemistry: Chemical Equilibrium and Le Chatelier's Principle
Predicting shifts in reversible reactions
Predicting shifts in reversible reactions
Chemistry - Grade 9-12
- 1
For the reaction N2(g) + 3H2(g) ⇌ 2NH3(g), write the equilibrium constant expression, Kc.
- 2
At equilibrium, the concentrations for H2(g) + I2(g) ⇌ 2HI(g) are [H2] = 0.20 M, [I2] = 0.20 M, and [HI] = 1.60 M. Calculate Kc.
- 3
For the equilibrium CO(g) + 2H2(g) ⇌ CH3OH(g), predict the shift if more CO is added to the container.
- 4
For the equilibrium 2SO2(g) + O2(g) ⇌ 2SO3(g), predict the shift if SO3 is removed from the container.
- 5
For the equilibrium N2O4(g) ⇌ 2NO2(g), the forward reaction is endothermic. Predict the shift when the temperature is increased.
- 6

For the equilibrium 2NO2(g) ⇌ N2O4(g), the forward reaction is exothermic. Predict the shift when the temperature is decreased.
- 7
For the equilibrium PCl5(g) ⇌ PCl3(g) + Cl2(g), predict the shift if the pressure is increased by decreasing the volume.
- 8
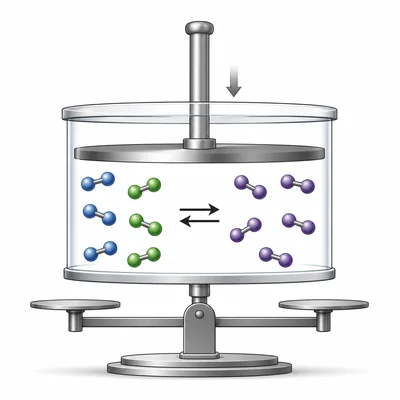
For the equilibrium H2(g) + Cl2(g) ⇌ 2HCl(g), predict the shift if the volume is decreased.
- 9

For the equilibrium CaCO3(s) ⇌ CaO(s) + CO2(g), write the equilibrium constant expression, Kc.
- 10
For the reaction 2A(g) ⇌ B(g), Kc = 25 at a certain temperature. If [A] = 0.50 M and [B] = 2.0 M, calculate Qc and determine whether the reaction will shift left, shift right, or remain at equilibrium.
- 11

For the equilibrium Fe3+(aq) + SCN-(aq) ⇌ FeSCN2+(aq), the product FeSCN2+ is deep red. Predict the color change when more SCN- is added.
- 12

For the equilibrium 4NH3(g) + 5O2(g) ⇌ 4NO(g) + 6H2O(g), predict the shift if pressure is increased by decreasing volume.
- 13
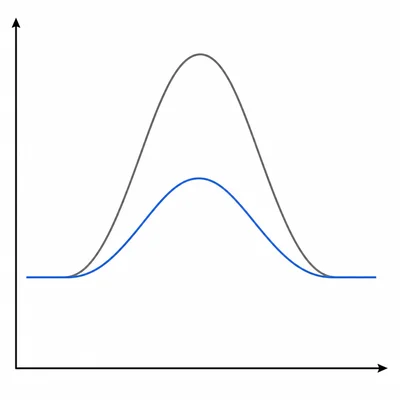
A catalyst is added to a reaction mixture already at equilibrium. Explain what happens to the position of equilibrium and to the rate at which equilibrium is reached.
- 14
For the equilibrium CO2(g) + H2(g) ⇌ CO(g) + H2O(g), Kc = 1.0 at a certain temperature. If the mixture contains [CO2] = 0.30 M, [H2] = 0.30 M, [CO] = 0.30 M, and [H2O] = 0.30 M, is the system at equilibrium?
- 15
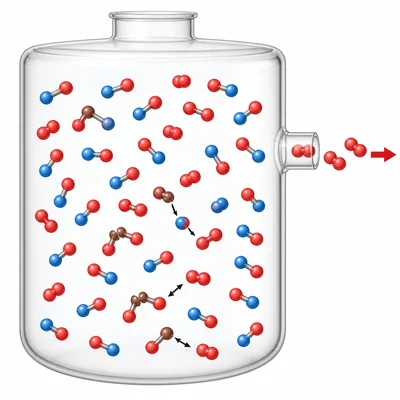
For the equilibrium 2NO(g) + O2(g) ⇌ 2NO2(g), predict the shift when O2 is removed and explain how the concentration of NO changes.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems